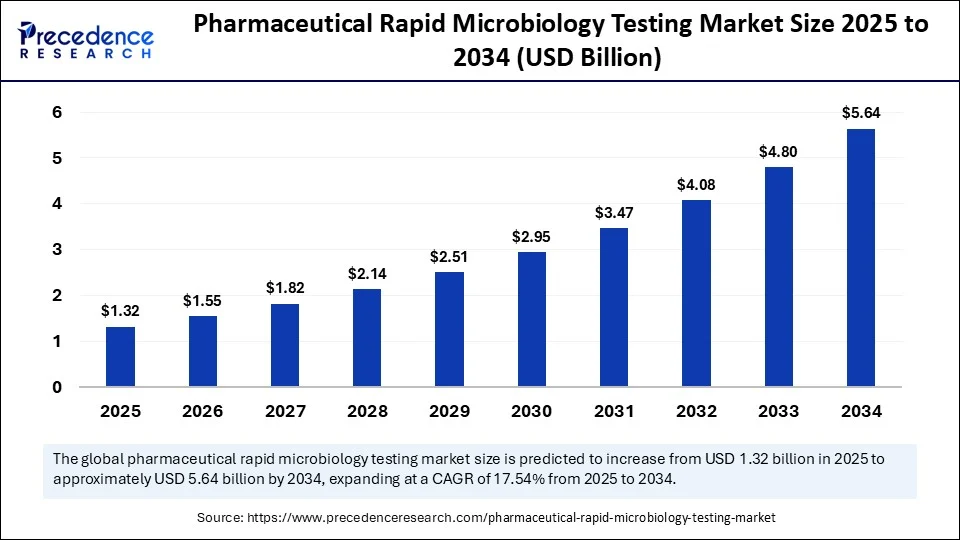
The pharmaceutical rapid microbiology testing market is projected to grow at a 17.54% CAGR, hitting USD 5.64B by 2034 from USD 1.12B in 2024.
Pharmaceutical Rapid Microbiology Testing Market Key Takeaways
- Global market set to grow from USD 1.12B in 2024 to USD 5.64B in 2034 at 17.54% CAGR.
- North America held the largest share (38%) in 2024; Asia Pacific shows fastest regional growth.
- PCR led technology share; flow cytometry will grow most rapidly.
- Product-wise, instruments dominate, while software & services see fastest gains.
- Leading application: biopharmaceuticals; fastest-growing: vaccines.
- Dominant microorganism: bacteria; viruses will grow fastest.
- Key end-user: pharma & biotech companies; CMOs emerging fast.
- Most common test: sterility testing; rising focus: environmental monitoring.
How is Artificial Intelligence Enhancing the Efficiency of Pharmaceutical Rapid Microbiology Testing?
Artificial intelligence is playing a pivotal role in revolutionizing pharmaceutical rapid microbiology testing (RMT). By accelerating data analysis and improving accuracy, AI minimizes both false positives and negatives in microbial detection. Its integration with automated systems enhances efficiency in processes like colony counting and sample handling. Additionally, AI-powered predictive models enable early detection of microbial risks, strengthening proactive quality control measures.
With the rise in sterile drug and biologics manufacturing, contamination concerns are intensifying. Consequently, advanced technologies such as biosensors, automation, and AI-based analytics are gaining traction—driving global adoption of RMT solutions focused on regulatory compliance and cost-effective quality assurance.
Get a Free Sample Copy of the Report@ https://www.precedenceresearch.com/sample/6332
Market Overview
The pharmaceutical rapid microbiology testing market is gaining momentum as pharmaceutical companies move away from conventional culture-based testing toward faster and more reliable solutions. This transition is primarily driven by the growing need for real-time quality control and the increasing complexity of drug manufacturing, particularly for biologics.
Drivers
The rise in sterile drug production, coupled with the enforcement of stricter microbial testing standards, is pushing pharma companies to adopt rapid microbiology tools. Technological advancements—such as AI integration, enhanced biosensors, and automated workflows—are also fueling growth by improving accuracy and throughput.
Opportunities
The market holds considerable potential in developing economies where pharmaceutical production is expanding rapidly. Increased investment in laboratory modernization and the growing role of CMOs in quality control processes create fertile ground for rapid microbiology testing solutions.
Challenges
Cost sensitivity in emerging markets and the capital investment required for advanced testing systems pose challenges to widespread adoption. Additionally, the lack of global standardization for validation methods and the training gap for specialized personnel limit the scalability of rapid testing solutions.
Regional Insights
North America leads the pharmaceutical rapid microbiology testing market due to its advanced pharmaceutical manufacturing ecosystem and proactive regulatory landscape. Meanwhile, Asia Pacific is witnessing accelerated growth, thanks to increased drug production and government initiatives to improve healthcare quality standards.
Pharmaceutical Rapid Microbiology Testing Market Companies

- Thermo Fisher Scientific Inc.
- Merck KGaA (MilliporeSigma)
- Bio-Rad Laboratories, Inc.
- Lonza Group AG
- Charles River Laboratories International, Inc.
- 3M Company
- Hamilton Company
- Microbiological Solutions (a part of Pall Corporation)
- Pall Corporation
- Pall Life Sciences
- Qiagen N.V.
- PerkinElmer, Inc.
- Eurofins Scientific
- IDEXX Laboratories, Inc.
- BioMérieux SA
- Neogen Corporation
- Agilent Technologies, Inc.
- Nova Biomedical Corporation
- LuminUltra Technologies Ltd.
- Accelerate Diagnostics, Inc.
Recent Developments
Major players in the market are introducing innovative products that integrate AI, robotics, and cloud analytics. Strategic partnerships and acquisitions have been seen in the space, focusing on expanding product portfolios and enhancing global presence. These developments aim to provide faster, more compliant, and scalable testing solutions across diverse production environments.
- On January 16, 2025, Rapid Infection Diagnostics Inc. (RID) introduced its BSIDx system, offering rapid pathogen identification and antibiotic sensitivity results for bloodstream infections in under five hours—cutting down the traditional processing time by approximately 30 hours.
- On March 4, 2025, Nelson Labs launched rapid sterility testing at three of its facilities—Salt Lake City and Itasca in the U.S., and Wiesbaden, Germany—significantly reducing incubation periods from 14 days to just six, all while remaining compliant with USP <71>.
- on April 22, 2025, Redberry successfully validated a four-day rapid sterility testing method for pharmaceutical products, minimizing standard release timelines and ensuring full microbial identification and regulatory adherence.
Segments Covered in the Report
By Technology Type
- Polymerase Chain Reaction (PCR)
- ATP Bioluminescence
- Flow Cytometry
- Enzyme-Linked Immunosorbent Assay (ELISA)
- Chromatography
- Biosensors & Bioassays
- Impedance Microbiology
- Others (e.g., Microfluidics, Microarray)
By Product Type
- Instruments
- Reagents & Consumables
- Software & Services
By Application
- Biopharmaceuticals
- Small Molecule Pharmaceuticals
- Vaccines
- Contract Research Organizations (CROs)
- Academic & Research Institutes
By Microorganism Type Tested
- Bacteria
- Fungi (Yeast and Mold)
- Viruses
- Mycoplasma
- Endotoxins
By End User
- Pharmaceutical & Biotechnology Companies
- Contract Manufacturing Organizations (CMOs)
- Hospitals & Diagnostic Laboratories
- Research & Academic Institutes
By Testing Type
- Sterility Testing
- Environmental Monitoring
- Bioburden Testing
- Raw Material Testing
- Water Testing
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
Ready for more? Dive into the full experience on our website!
