Bimekizumab Market Key Takeaways
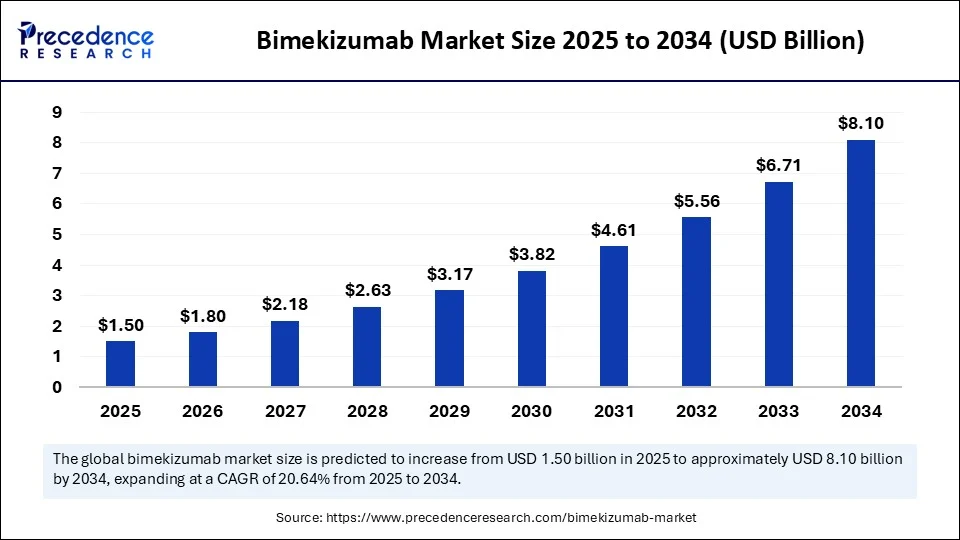
- In terms of revenue, the global bimekizumab (BIMZELX) market was valued at USD 1.24 billion in 2024.
- It is projected to reach USD 8.10 billion by 2034.
- The market is expected to grow at a CAGR of 20.64% from 2025 to 2034.
- North America dominated the market with the largest market share of 39% in 2024.
- Asia Pacific is expected to expand at the fastest CAGR between 2025 and 2034.
- By indication, the plaque psoriasis (PSO) segment captured the biggest market share in 2024.
- By indication, the hidradenitis suppurativa (HS) segment is expected to grow at the fastest CAGR between 2025 and 2034.
- By formulation, the pre-filled syringe (160 mg / 1 mL) segment contributed the highest market share in 2024.
- By formulation, the pre-filled syringe or pen (320 mg / 2 mL) segment is expected to grow at the highest CAGR between 2025 and 2034.
- By distribution channel, the hospital pharmacies segment generated the major market share in 2024.
- By distribution channel, the online pharmacies segment is expected to grow at a significant CAGR between 2025 and 2034.
How Is AI Reshaping the Bimekizumab Market?
Artificial intelligence is emerging as a key enabler in the expansion and optimization of the Bimekizumab (BIMZELX) market, influencing nearly every stage of its clinical and commercial journey. AI-driven tools are enhancing early diagnosis by streamlining patient screening, allowing healthcare providers to efficiently identify candidates who are most likely to benefit from BIMZELX.
Predictive analytics are helping determine likely responders, enabling more precise treatment planning and improving long-term patient adherence. In clinical development, AI is accelerating trial design, optimizing patient recruitment, and reducing time-to-market for future label expansions.
Pharmaceutical companies are also leveraging AI-based sentiment analysis to better understand patient and physician behavior, which helps refine marketing strategies and enhance engagement with prescribers. On the operational side, AI-powered supply chain platforms are improving the distribution and availability of BIMZELX across specialty pharmacy networks.
Altogether, AI is not only contributing to market growth but is fundamentally reshaping how advanced immunological therapies like BIMZELX are developed, accessed, and managed in real-world settings.
Market Overview
The bimekizumab (BIMZELX) market encompasses the commercial landscape surrounding UCB’s dual IL-17A and IL-17F inhibitor monoclonal antibody, bimekizumabbkzx. The market includes all approved indications—such as plaque psoriasis (PSO), psoriatic arthritis (PsA), ankylosing spondylitis (AS), non-radiographic axial spondyloarthritis (nr-axSpA), and hidradenitis suppurativa (HS) along with its formulation types, distribution pathways, and regional uptake trends projected through 2034.
As of 2025, the market is experiencing strong growth, particularly within dermatology and rheumatology segments. Bimekizumab’s unique dual inhibition mechanism distinguishes it from IL-17A-only therapies, offering enhanced clinical benefits including faster skin clearance and greater durability of response. These advantages have contributed to a robust uptake in key regions, further supported by favorable reimbursement environments and growing acceptance of advanced biologics.
The global footprint of BIMZELX continues to expand as more emerging markets adopt targeted immunotherapies. Although competition from established biologics like Cosentyx and Taltz remains significant, bimekizumab’s clinical differentiation provides a competitive advantage. In addition, ongoing late-stage clinical trials for new indications, including hidradenitis suppurativa, are expected to strengthen its market position further. Overall, the outlook for the BIMZELX market remains positive, driven by innovation, expanding access, and continued clinical success.
Market Trends
- Rising preference for dual-cytokine inhibition therapies due to faster disease control
- Increased demand for treatments with rapid onset of action
- Growing focus on sustained disease management in therapeutic choices
- Enhanced patient and physician emphasis on treatment experience and outcomes
- Rising demand for personalized and combination therapies
Opportunity
Expanding Indications and Combination Therapies
The next wave of market growth lies in exploring the efficacy of bimekizumab beyond plaque psoriasis. Ongoing studies in conditions like hidradenitis suppurativa and axial spondylarthritis present sizable future opportunities. Additionally, pediatric trials and use in underserved populations open new access points. As biosimilar pressure on older biologics, bimekizumab can position itself as a next-generation innovation. Exploring the use of bimekizumab in combination therapies further opens up new avenues for growth.
Recent Development
- In February 2025, UCB’s dual IL 17A/IL 17F inhibitor, Bimzelx (bimekizumab), demonstrated strong early momentum following its launch in the U.S. psoriatic arthritis (PsA) market. A Spherix Global Insights survey conducted within three months of launch revealed that rheumatologists are embracing Bimzelx more rapidly and with greater enthusiasm than many other recently introduced biologics.
Bimekizumab Market Top Companies

