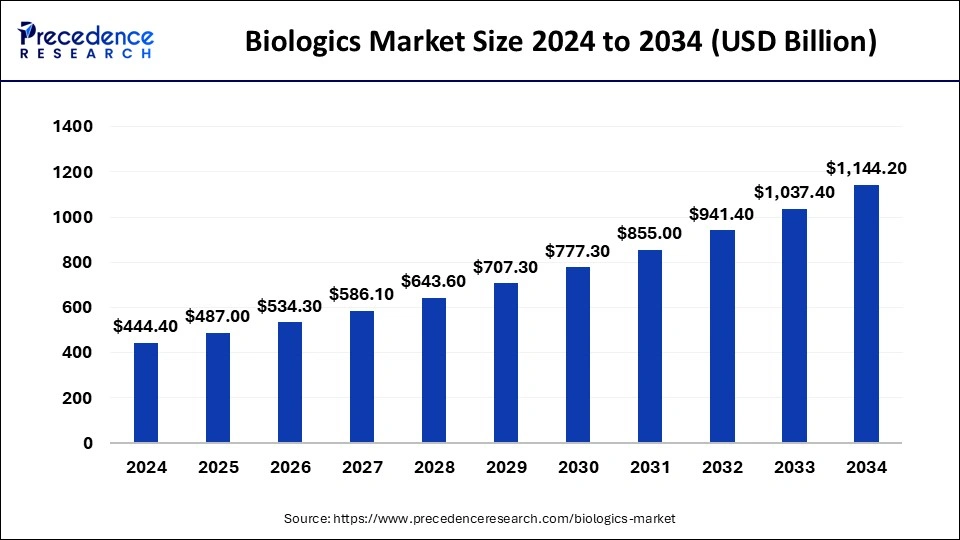
Biologics Market Key Takeaways
- North America contributed more than 44.53% in 2024.
- Asia Pacific region is projected to expand at the CAGR of 11.05% during the forecast period.
- By source, the microbial segment dominated the market with a share of 58.78% in 2024.
- By manufacturing, the in-house segment dominated the market with 83.96% share in 2024.
- By product, the monoclonal antibodies segment accounted for a share of 56.48% in 2024.
Biologics Market Growth Factors
Biologics are therapeutic substances derived from living organisms or their components, and include a broad category of biotechnology-based pharmaceuticals developed from humans, animals, and microorganisms. These therapies may consist of proteins that regulate cellular functions, genes that influence protein synthesis, modified hormones, and cells that activate or suppress immune responses. Due to their ability to modulate biological pathways, biologic therapies are often referred to as biologic response modifiers.
The increasing prevalence and improved diagnosis of chronic diseases have driven demand for advanced diagnostic tools and targeted treatments, significantly boosting the global biologics market. Biologic drugs are specifically engineered to interact with precise components of the immune system responsible for inflammation and disease progression. In the wake of global health challenges like COVID-19, governments have enhanced support for healthcare infrastructure and prioritized the production and distribution of biologics to strengthen public health preparedness.
Additionally, ongoing research in species and expression systems aims to increase the yield and efficiency of biologic production. Pharmaceutical companies are heavily investing in R&D to enhance the efficacy of biologics, particularly in the development of oral biologic options for conditions like arthritis, which could improve accessibility and patient adherence.
As productivity in small molecule drug development continues to decline, biologics are expected to play a more dominant role in the pharmaceutical landscape. Companies are expanding their biologic pipelines to maintain competitive advantage. Innovations in therapies for conditions like Crohn’s disease and rheumatoid arthritis are also encouraging a shift among moderately affected patients from traditional oral medications to novel biologic treatments.
Furthermore, emerging biologic compounds provide alternative treatment options for patients unresponsive to conventional therapies. These newer agents often demonstrate improved safety profiles and greater therapeutic efficacy, positioning them as valuable tools in personalized and precision medicine.
Opportunity
Rising Demand for Personalized Medicine
The growing emphasis on personalized medicine is creating significant opportunities for the biologics market. Personalized medicine relies on targeted therapies that address specific molecular pathways and genetic profiles. Biologics play a critical role in this space by enabling the development of precision diagnostic tools, such as biomarkers, which help identify disease subtypes and guide tailored treatment approaches.
As healthcare increasingly shifts toward individualized care, there is a rising demand for therapies that can be customized to a patient’s unique biological makeup. Biologics, due to their adaptability and specificity, can be engineered to meet these personalized needs, resulting in more effective and patient-centric treatment outcomes. This trend is expected to drive innovation and investment in biologic drug development, expanding the market’s potential across various therapeutic areas.
Recent Developments
- Moderna Inc., announced in August 2021 that it had completed the rolling submission process for its Biologics License Application to the FDA for full licensure of the Moderna COVID-19 vaccine for active immunization to prevent COVID-19 in people aged 18 and up.
- Pfizer Inc. and BioNTech SE declared in August 2021 that they had started a supplemental Biologics License Application with the FDA for the approval of a booster dose of COMIRNATY to prevent COVID-19 in people aged 16 and up.
- As India’s biggest biosimilar maker increases its production to generate value to stakeholders ahead of an anticipated initial pubic offering, Biocon Biologics is in talks with ADQ, the Abu Dhabi government’s wealth fund, to advance capital.
- A South Korean business, Samsung Biologics has committed to work with Eli Lilly & Company on the development of a virus neutralizing antibody for the COVID-19 virus.
- In 2020, WuXi Biologics completed purchase of Bayer’s drug product manufacturing facility in Germany. WuXi Biologics’ first medication production facility in Europe was expected to increase the company’s commercial manufacturing capabilities.
- In October 2020, Cadila Pharmaceuticals released two similar biologics known as NuPTH and Cadalimab in the Indian market to increase its geographical reach.
Segments Covered in the Report
By Source
- Microbial
- Mammalian
- Others
By Product
- Monoclonal Antibodies
- Human mABs
- Humanized mABs
- Chimeric mABs
- Murine mABs
- Vaccines
- RecombinantProteins
- Antisense, RNAi & molecular therapy
- Cell Based Therapies
- Stem Cell Therapy
- CAR-T Cell Therapy
- Tissue Engineering
- Others
By Indication
- Oncology
- Immunological Disorders
- Cardiovascular Disorders
- Hematological Disorders
- Others
By Manufacturing
- Outsourced
- In-house
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
By Geography
-
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
