GI Stool Testing Market Key Takeaways
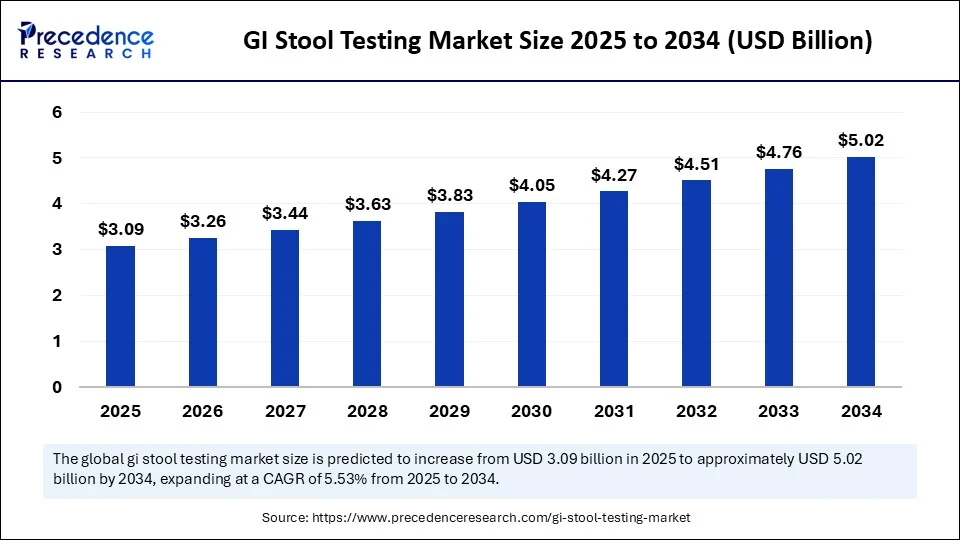
- In terms of revenue, the global GI stool testing market was valued at USD 2.93 billion in 2024.
- It is projected to reach USD 5.02 billion by 2034.
- The market is expected to grow at a CAGR of 5.53% from 2025 to 2034.
- North America dominated the GI stool testing market with the largest market share of 41.6% in 2024.
- Asia Pacific is expected to expand at the fastest CAGR between 2025 and 2034.
- By test type, the occult blood test (FOBT, FIT) dominated the market, under which the immunochemical-based FOBT (iFOBT or FIT) sub-segment held a 22.5%
- market share in 2024.
- By test type, the gut microbiome testing segment is anticipated to grow at a remarkable CAGR between 2025 and 2034.
- By technology, the immunoassay segment captured the biggest market share of 26.4% in 2024.
- By technology, the next generation sequencing (NGS) segment is expected to expand at a notable CAGR over the projected period.
- By application, the colorectal cancer screening segment held the highest market share of 33.2% in 2024.
- By application, the gut microbiome health assessment segment is anticipated to grow at a remarkable CAGR between 2025 and 2034.
- By sample collection, the hospital / lab collection segment captured the major market share of 57.8% in 2024.
- By sample collection, the at-home sample collection kits segment is expected to expand at a notable CAGR over the projected period.
- By end user, the hospitals & clinics segment generated the remarkable market share of 38.1% in 2024.
- By end user, the homecare / individual users segment is expected to expand at a notable CAGR over the projected period.
How AI is Improving Diagnostic Accuracy and Expanding Access to GI Stool Testing
Artificial intelligence is revolutionizing gastrointestinal (GI) stool testing by making it more accurate, accessible, and non-invasive. AI-powered algorithms can analyze large volumes of test data, delivering personalized health insights and early alerts based on stool samples submitted through at-home kits. This data-driven approach is transforming GI diagnostics into a more proactive and patient-friendly process.
In Singapore, researchers have developed a deep learning model capable of detecting traces of blood in stool using images captured by a smartphone. The model, enhanced with Generative Adversarial Networks (GANs), powers an app called “Poolice,” which demonstrated 94% accuracy in identifying potential signs of gastrointestinal bleeding during trials.
Meanwhile, U.S.-based biotech company Viome has distributed over 500,000 AI-enabled stool testing kits for at-home use. These kits leverage RNA and metatranscriptomic analysis to provide users with detailed insights into gut health, further demonstrating how AI is broadening access and enhancing the accuracy of GI diagnostics.
Market Overview
The gastrointestinal (GI) stool testing market involves a range of diagnostic tests conducted on fecal samples to detect various gastrointestinal conditions, infections, inflammation, and imbalances in the gut microbiome. These tests play a vital role in identifying disorders such as colorectal cancer, Clostridium difficile infection, inflammatory bowel disease (IBD), irritable bowel syndrome (IBS), parasitic infections, and other digestive system abnormalities.
The market includes a variety of testing methods, such as traditional laboratory diagnostics, point-of-care (PoC) testing, and advanced molecular-based stool testing kits. These tests cover a broad spectrum of diagnostic markers, including occult blood, bacterial, viral, and parasitic pathogens, inflammatory biomarkers like calprotectin and lactoferrin, digestive enzymes, and comprehensive microbiome profiling.
As awareness of gut health and early disease detection grows, the demand for non-invasive, accurate, and convenient stool testing solutions is driving innovation and adoption across both clinical and home-based diagnostic settings.
Key Growth Factors of the GI Stool Testing Market
- Rising Incidence of Gastrointestinal Disorders: The growing prevalence of conditions such as gastrointestinal infections, inflammatory bowel disease (IBD), and colorectal cancer is driving demand for accurate and timely stool-based diagnostics. These tests support early detection and intervention, improving patient outcomes.
- Advancements in Diagnostic Technologies: The development of molecular diagnostics, including multiplex PCR and next-generation sequencing, has significantly enhanced the speed, accuracy, and comprehensiveness of stool testing. These innovations are improving the detection of a wide range of pathogens and biomarkers in both acute and chronic GI conditions.
- Growing Emphasis on Preventive Healthcare: Increased public awareness around the importance of early disease screening is fueling the adoption of stool tests as a non-invasive method to detect latent gastrointestinal issues before symptoms arise, contributing to preventive care strategies.
- Aging Population and Chronic Disease Burden: As the global population ages, the incidence of age-related gastrointestinal disorders continues to rise. Older adults increasingly rely on stool tests for routine monitoring and disease management due to their convenience and non-invasive nature.
- Supportive Health Policies and Screening Programs: Government-backed screening initiatives and expanded insurance coverage for non-invasive stool tests, particularly for colorectal cancer screening, are improving accessibility and encouraging higher rates of compliance. These policy measures are vital in expanding the market, especially in population-wide preventive health programs.
Opportunity
The Rise of At-Home RNA-Based Stool Tests
A major future opportunity for the GI stool testing market lies in the emergence of advanced, at-home RNA-based diagnostic tests that provide non-invasive, highly sensitive detection for gastrointestinal conditions, particularly colorectal cancer. In May 2024, the U.S. FDA approved ColoSense, the first multitarget stool RNA (mt-sRNA) test for adults over 45 for colorectal cancer screening. The test, developed by Geneoscopy, demonstrated 94.4% sensitivity in clinical study reports and is designed for at-home self-collection of stool samples with medical confirmation if positive.
This initial approval represents a shift away from traditional diagnostics to patient-centered remote diagnostics and testing, particularly for the early diagnostic disease continuum. Governments and health insurers are increasingly supporting the use of mail-in sample kits to provide easier access for patients to increase screening rates and reduce reliance on hospital visits. With the rise in GI diseases and colorectal cancer worldwide, the incorporation of at-home RNA-based stool tests may provide new options for early intervention, development of a public health approach, and industry expansion, making them potentially one of the most attractive growth drivers in the industry.
GI Stool Testing Market Key Players

- Exact Sciences Corporation
- BioFire Diagnostics (bioMérieux)
- QuidelOrtho Corporation
- Abbott Laboratories
- Thermo Fisher Scientific Inc.
- Becton, Dickinson and Company
- Roche Diagnostics
- Hologic, Inc.
- CerTest Biotec
- Alere Inc. (now part of Abbott)
- Genova Diagnostics
- Diagnostic Solutions Laboratory
- Promega Corporation
- Biomerica, Inc.
- Gastrointestinal Diagnostic Laboratory (GDX)
- Biomerieux SA
- Siemens Healthineers
- MyBioma
- DayTwo
- Seegene Inc.
Recent Development
- In August 2024, MP Biomedicals launched advanced gastrointestinal diseases diagnostic kits. This test kit includes Rapid H.pylori Antigen Test Card is an in-vitro qualitative immunochromatographic assay for the rapid detection of Helicobacter pylori antigens in human stool specimen.
- In November 2024, EmeritusDX, announced the launch of GI Detect™, a laboratory diagnostic test designed to diagnose gastrointestinal disturbances. This test utilizes a single stool swab to identify 26 bacteria, viruses, and parasites known to disrupt the digestive system.
Segments Covered in the Report
By Test Type
- Occult Blood Test (FOBT, FIT)
- Guaiac-based FOBT (gFOBT)
- Immunochemical-based FOBT (iFOBT or FIT)
- Microbial Testing
- Bacterial Infections
- Viral Infections
- Parasitic Infections
- Calprotectin & Inflammatory Markers
- Calprotectin Test
- Lactoferrin Test
- M2-PK (M2 Pyruvate Kinase)
- Digestive Function Tests
- Pancreatic Elastase
- Fecal Fat Test
- Molecular Diagnostics
- PCR-based Panels
- NGS (Next Generation Sequencing)
- Gut Microbiome Testing
- Targeted Bacterial Testing
- Comprehensive Microbiota Profiling
- Others
- pH Testing
- Reducing Substances
- Alpha-1 Antitrypsin
By Technology
- Immunoassay
- Lateral Flow Assay (LFA)
- Enzyme-Linked Immunosorbent Assay (ELISA)
- Polymerase Chain Reaction (PCR)
- Next Generation Sequencing (NGS)
- Mass Spectrometry
- Others
By Application
- Infectious Gastroenteritis
- Colorectal Cancer Screening
- Inflammatory Bowel Disease (IBD)
- Irritable Bowel Syndrome (IBS)
- Nutritional Malabsorption Disorders
- Gut Microbiome Health Assessment
- Others
By Sample Collection Method
- At-home Sample Collection Kits
- Hospital / Laboratory Collection
- Others
By End User
- Hospitals & Clinics
- Diagnostic Laboratories
- Academic & Research Institutions
- Homecare / Individual Users
- Others
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
