Pentoxifylline Drug Market Key Takeaways
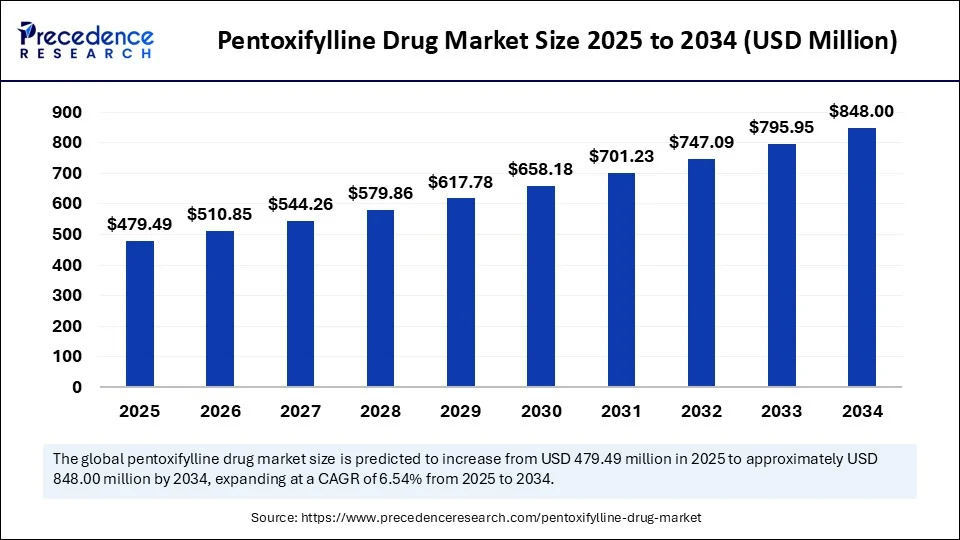
- In terms of revenue, the global pentoxifylline drug market was valued at USD 450.06 million in 2024.
- It is projected to reach USD 848.00 million by 2034.
- The market is expected to grow at a CAGR of 6.54% from 2025 to 2034.
- North America dominated the global market with the largest market share of 35% in 2024.
- Asia Pacific is anticipated to grow at a significant CAGR from 2025 to 2034.
- By dosage form, the tablet segment held the biggest market share of 60% in 2024.
- By dosage form, the injection segment is expected to grow at the fastest CAGR during the foreseeable period.
- By route of administration, the oral segment captured the highest market share of 70% in 2024.
- By route of administration, the IV/injectable segment is anticipated to grow at a significant CAGR from 2025 to 2034.
- By application/indication, the peripheral vascular disease (PVD) segment contributed the major market share of 40% in 2024.
- By application/indication, the diabetic neuropathy segment is expanding at a significant CAGR.
- By distribution channel, the hospital pharmacies segment held the largest market share of 45% in 2024.
- By distribution channel, the online pharmacies segment is projected to grow at a notable CAGR between 2025 and 2034.
AI Impact on the Pentoxifylline Drug Market
Artificial Intelligence (AI) is reshaping the pentoxifylline drug market by accelerating drug discovery, optimizing clinical trials, and improving treatment outcomes. Through advanced algorithms, AI can process vast datasets to identify novel drug targets, predict efficacy and toxicity, and support the development of personalized treatment strategies. For pentoxifylline specifically, AI models help forecast pharmacokinetic properties, efficacy levels, and potential side effects, reducing reliance on lengthy and costly laboratory testing. Moreover, AI-driven analysis of genomic and proteomic data enables researchers to uncover new therapeutic applications or develop improved formulations of pentoxifylline, broadening its clinical potential and market reach.
Market Overview
Pentoxifylline, a xanthine derivative, is widely prescribed to improve blood flow in patients suffering from peripheral vascular diseases, chronic occlusive arterial disease, and intermittent claudication. The drug works by decreasing blood viscosity, enhancing red blood cell flexibility, and reducing platelet aggregation. The pentoxifylline drug market encompasses the production, distribution, and commercialization of pharmaceutical formulations used in vascular disorders, neurological conditions, and various off-label applications. Market expansion is fueled by the increasing prevalence of cardiovascular and peripheral vascular diseases, the rapidly growing geriatric population, and the rising adoption of both oral and injectable formulations across hospitals, clinics, and outpatient care settings.
Key Trends in the Pentoxifylline Drug Market
- Growing Awareness and Expanded Healthcare Access: Rising awareness of vascular diseases, coupled with better healthcare infrastructure in developing countries, is increasing the patient pool eligible for pentoxifylline treatment.
- Innovations in Drug Delivery Systems: Ongoing research into advanced delivery mechanisms, including sustained-release formulations, is aimed at improving therapeutic efficacy, enhancing patient compliance, and reducing dosing frequency.
- Affordability and Widespread Availability: Generic versions of pentoxifylline provide a cost-effective alternative to newer, more expensive therapies. This affordability is particularly valuable in price-sensitive regions and developing economies where access to healthcare is steadily improving.
- Supportive Regulations and Clinical Validation: A growing base of clinical evidence, combined with favorable regulatory policies in several regions, is driving physician confidence and supporting the broader adoption of pentoxifylline in clinical practice.
Drivers
Increasing Prevalence of Peripheral Arterial Disease (PAD)
The growing incidence of peripheral arterial disease (PAD) and other chronic vascular complications, particularly among the elderly and diabetic population, is a major factor driving market growth. Pentoxifylline plays a vital role in treating intermittent claudication by reducing blood viscosity, improving red blood cell flexibility, and enhancing circulation. Its therapeutic value is further supported by ongoing research into broader applications, such as diabetic kidney disease and immunomodulation, making it a versatile option for multiple vascular and inflammatory conditions.
Restraints
Competition from Generics and Alternative Therapies
The pentoxifylline market is facing downward pressure due to the availability of low-cost generics, leading to significant price competition and reduced profitability for branded formulations. In addition, the emergence of newer therapies with higher efficacy and fewer side effects poses a challenge to pentoxifylline’s clinical relevance. Safety concerns and reported side effects, along with strict regulatory requirements for new formulations and evolving treatment guidelines, further restrict market expansion.
Opportunities
Expanding Applications in New Indications
The greatest opportunity for market growth lies in extending pentoxifylline’s therapeutic use beyond its established role in intermittent claudication. Research highlights its potential in treating diabetic kidney disease (DKD), where its anti-inflammatory and anti-fibrotic properties may reduce proteinuria and slow kidney function decline. Exploring additional applications in nephrology, neurology, and immunology could create new revenue streams, especially if supported by favorable clinical trial outcomes and regulatory approvals.
Recent Developments
- In December 2024, Adalvo launched the first Lisdexamfetamine Oral Solution in Germany, developed in partnership and patented in Europe. This child-friendly formulation treats attention deficit hyperactivity disorder (ADHD) and follows the revocation of the supplementary protection certificate (SPC) by the Federal Patent Court. Adalvo’s abbreviated new drug application (ANDA) for the solution was also accepted by the U.S. FDA, ensuring high-quality and timely product launches.
- In January 2024, ANI Pharmaceuticals launched Pentoxifylline Extended-Release Tablets, a generic version of Trental®, targeting a U.S. market worth USD 19.7 million. ANI emphasizes growth through new product launches and reliable supply, focusing on patient access to quality therapeutics to ensure that both patients and healthcare providers have access to high-quality medications.
Pentoxifylline Drug Market Key Players

- Sanofi S.A.
- Mylan N.V. (Viatris)
- Teva Pharmaceuticals
- Dr. Reddy’s Laboratories
- Cipla Limited
- Sun Pharmaceutical Industries
- Glenmark Pharmaceuticals
- Lupin Limited
- Aurobindo Pharma
- Zydus Cadila
- F. Hoffmann-La Roche Ltd
- Sandoz International GmbH
- Torrent Pharmaceuticals
- Medopharm
- Pharmstandard
- Hikma Pharmaceuticals
- Laboratoires Servier
- Natco Pharma
- Alembic Pharmaceuticals
- Intas Pharmaceuticals
