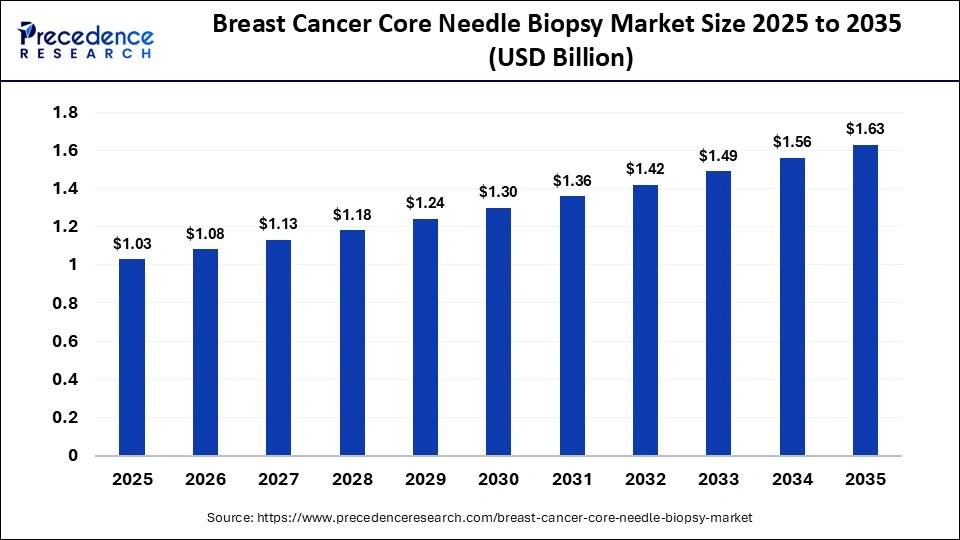
Market Key Takeaways
- North America led the breast cancer core needle market and accounted for the largest share of 46.70% in 2025.
- The Asia Pacific is estimated to grow at a CAGR of 5.37% between 2026 and 2035.
- By technology, the ultrasound-based breast biopsy segment captured the biggest market share of 42% in 2025.
- By technology, the CT-based breast biopsy segment is expected to expand with the highest CAGR over the projected period.
- By end-user, the hospitals and diagnostic laboratories companies segment held a dominant market share of 61% in 2024.
- By end-user, the pharmaceutical & biotechnology companies segment is expected to expand at the fastest CAGR from 2026 to 2034.
How Is Breast Cancer Core Needle Biopsy Demand Motivated?
Demand for breast cancer core needle biopsy is being driven by the growing emphasis on early, accurate, and minimally invasive cancer diagnosis. Core needle biopsy allows clinicians to confirm breast cancer without the need for surgical procedures, offering high diagnostic accuracy with reduced patient discomfort, faster recovery, and lower overall costs. As the global incidence of breast cancer continues to rise, healthcare systems are prioritizing efficient diagnostic methods that support early detection and timely treatment.
The expansion of population-wide screening programs and increased awareness of breast cancer have significantly increased the number of diagnostic procedures performed in hospitals and specialized diagnostic centers. Technological advancements particularly in imaging-guided biopsy techniques such as ultrasound-, mammography-, and MRI-guided systems are improving sampling precision and reducing the need for repeat procedures. In addition, patient preference for minimally invasive diagnostic options, combined with government initiatives promoting early cancer detection, is further supporting market growth. Increased investment by diagnostic and medical device companies, along with expanding access to screening services in underserved and emerging regions, is accelerating adoption and driving sustained demand for breast cancer core needle biopsy solutions.
Will AI Revolutionise Core-Needle Breast Biopsy Diagnostics?
Artificial intelligence is increasingly transforming breast core-needle biopsy (CNB) diagnostics by enhancing histopathological interpretation, imaging assessment, and risk stratification. AI models applied to whole-slide pathology images and molecular profiles can achieve high sensitivity with low false-negative rates, helping pathologists identify malignant features more efficiently and prioritize biopsy cores for review. This has significantly shortened the time from tissue sampling to diagnostic reporting, improving clinical decision-making and workflow efficiency.
Recent multicentre studies demonstrate AI’s capability to assess the likelihood of disease upstaging from ductal carcinoma in situ (DCIS) to invasive breast cancer, supporting more informed surgical planning and treatment selection. AI systems are also showing promise in identifying lesions with aggressive progression potential, guiding neoadjuvant therapy decisions, and reducing unnecessary repeat procedures. Large-scale validation efforts, such as the NHS’s AI trials in mammography and regional deployments in other countries, highlight accelerating clinical adoption and confidence in AI-driven diagnostic tools.
In underserved and resource-limited settings, AI-enabled CNB diagnostics offer the potential to improve access to expert interpretation, enable remote second opinions, and reduce diagnostic disparities. However, widespread implementation still faces challenges related to regulatory approval, data standardization, and model transparency. Despite these hurdles, ongoing validation and integration efforts suggest AI is poised to play a transformative role in advancing accuracy, efficiency, and equity in breast core-needle biopsy diagnostics.
Growth Factors of the Breast Cancer Core Needle Biopsy Market
- Rising Incidence of Breast Cancer: Increasing global prevalence of breast cancer is driving demand for accurate, reliable, and minimally invasive diagnostic procedures such as core needle biopsy.
- Emphasis on Early and Accurate Diagnosis: Healthcare systems are prioritizing early detection to improve survival rates, fueling adoption of core needle biopsy as a first-line diagnostic tool.
- Growth of Breast Cancer Screening Programs: Expansion of population-based mammography and screening initiatives is increasing the number of suspicious lesions requiring biopsy confirmation.
- Technological Advancements in Imaging-Guided Biopsy: Improvements in ultrasound-, stereotactic-, and MRI-guided biopsy systems are enhancing accuracy, reducing repeat procedures, and improving patient outcomes.
- Patient Preference for Minimally Invasive Procedures: Core needle biopsy offers reduced pain, faster recovery, and lower costs compared to surgical biopsy, supporting wider acceptance.
- Integration of AI and Digital Pathology: AI-assisted image analysis and pathology interpretation are improving diagnostic confidence, turnaround times, and workflow efficiency.
- Increased Investment in Oncology Diagnostics: Medical device manufacturers and diagnostic companies are expanding portfolios and investing in innovation, supporting market expansion.
Regional Insights of the Breast Cancer Core Needle Biopsy Market
North America leads the market due to advanced healthcare infrastructure, high awareness of breast cancer screening, and early adoption of minimally invasive diagnostic technologies. The United States plays a key role, supported by strong reimbursement frameworks, widespread availability of imaging-guided biopsy systems, and growing integration of AI in pathology and radiology workflows.
Europe represents a well-established market, driven by national breast cancer screening programs and strong public healthcare systems. Countries such as the UK, Germany, France, and the Nordic region are witnessing steady growth, supported by technological advancements and increasing use of AI in diagnostics. Large-scale clinical validation initiatives across Europe are further strengthening adoption.
Asia Pacific is the fastest-growing region, fueled by rising breast cancer incidence, expanding healthcare access, and increasing investments in diagnostic infrastructure. Countries including China, India, Japan, and South Korea are expanding screening coverage and adopting cost-effective, minimally invasive biopsy techniques to meet growing demand.
Latin America is experiencing gradual market growth as awareness of early cancer detection improves and healthcare systems expand diagnostic capabilities. Brazil and Mexico are key contributors, supported by increasing public–private healthcare investments and access to imaging technologies.
The Middle East & Africa region is emerging, with growth driven by improving healthcare infrastructure, government-led cancer awareness programs, and increasing availability of diagnostic services. While adoption rates vary, ongoing investments in oncology care and telepathology solutions are expected to support long-term market development.
Breast Cancer Core Needle Biopsy Market Companies
- Intact Medical Corporation
- Ethicon Surgical Technologies
- Gallini SRL
- Leica Biosystems Nussloch GmbH
- Hologic, Inc.
- Argon Medical Devices
- Encapsule Medical Devices LLC
- Cook Medical Incorporated
- Becton, Dickinson and Company
- C.R. Bard, Inc.
Recent Developments
- In July 2025, researchers from National Taiwan University developed a miniature mass spectrometry platform coupled with paper spray ionization (MiniMaP) for rapid breast cancer diagnosis.
- In October 2025, Olympus announced the launch of SecureFlex, a single-use fine needle biopsy device for endoscopic ultrasound-guided procedures, improving tissue sampling for pancreatic and other cancer diagnoses, available first in Europe and Japan
- 10 September 2025, researchers developed CoreView imaging on needle, a low-cost system for rapid point-of-care imaging of breast core-needle biopsy samples, producing near histology-grade images within minutes to improve diagnostics in low-resource settings
Segments Covered in the Report
By Technology
- MRI-based Breast Biopsy
- Ultrasound-based Breast Biopsy
- Mammography-based Breast Biopsy
- CT-based Breast Biopsy
- Other Image-Based Breast Biopsy
By End-use
- Hospitals & Diagnostic Laboratories
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Get Sample Link : https://www.precedenceresearch.com/sample/7239
