Market Key Takeaways
- North America led the global market, holding more than 38% of market share in 2025.
- The Asia Pacific is estimated to expand the fastest CAGR between 2026 and 2035.
- By omics type, the genomic segment contributed the biggest market share in 2025.
- By omics type, the multi-omics segment is growing at a strong CAGR between 2026 and 2035.
- By study phase, the phase II segment dominated and held the biggest market share in 2025.
- By study phase, the phase I segment is expected to grow at the fastest CAGR from 2026 to 2035.
- By application, the oncology segment captured the highest market share in 2025.
- By application, the rare & genetic diseases segment is expanding at a significant CAGR between 2026 and 2035.
- By end-user, the pharmaceutical companies segment dominated the market in 2025.
- By end-user, the biotechnology companies segment is poised to grow at the fastest CAGR between 2026 and 2035.
- By service type, the omics data generation segment held the major market share in 2025.
- By service type, the bioinformatics & data analytics segment is expected to expand at the highest CAGR between 2026 and 2035.
Can Omics Transform the Way Clinical Trials Are Conducted?
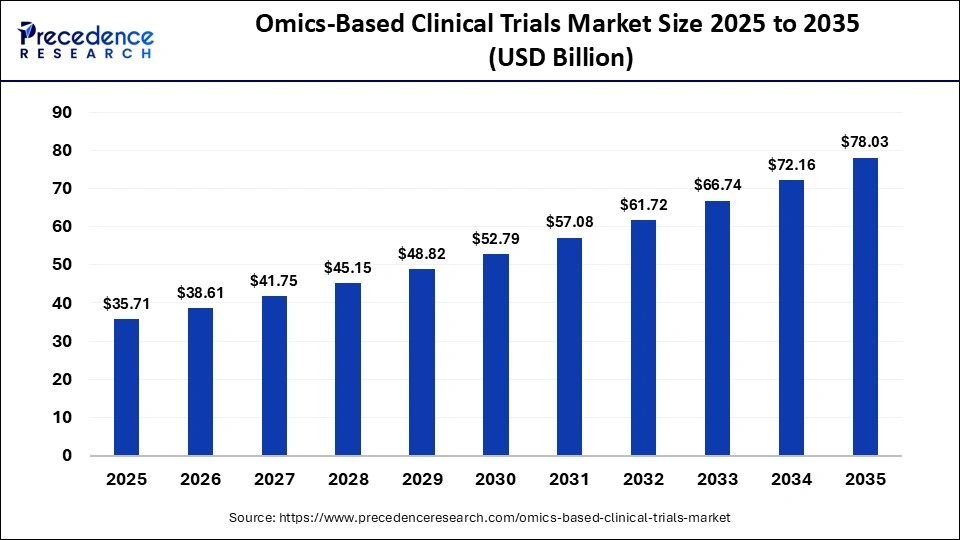
The omics-based clinical trials market is reshaping clinical research by integrating genomics, proteomics, transcriptomics, and metabolomics to enable more precise and data-driven trial designs. These technologies generate high-throughput biological insights that support accurate patient stratification, predictive response analysis, and outcome optimization, forming the foundation of precision medicine–focused clinical trials.
As adoption increases, drug developers are shifting toward biomarker-driven and targeted therapy trials to improve success rates and reduce development timelines. Advances in next-generation sequencing (NGS), multi-omics integration platforms, and sophisticated bioinformatics tools have significantly enhanced data accuracy, depth, and clinical applicability. In parallel, growing collaboration among contract research organizations (CROs), technology providers, and academic research institutions is accelerating the implementation of omics-enabled trial models. Moving forward, the ability to effectively integrate, analyze, and interpret complex multi-omics datasets will be a critical differentiator and a key driver of competitive advantage in this rapidly evolving market.
How Artificial Intelligence Is Transforming Omics-Based Clinical Trials in Precision Medicine
Artificial intelligence is fundamentally reshaping omics-based clinical trials by enabling the integration and advanced analysis of massive genomic, proteomic, transcriptomic, and multi-omic datasets. AI-driven analytics help researchers uncover previously unrecognized biomarkers, improve disease stratification, and generate deeper biological insights that support truly personalized therapeutic approaches. By automating complex multi-omics data processing and supporting adaptive trial designs, AI significantly enhances patient selection accuracy and optimizes trial efficiency.
Advanced genomic AI tools are now capable of identifying trial-eligible patients from highly complex datasets, reducing recruitment time while improving clinical relevance. In January 2025, NVIDIA partnered with Illumina to combine genomics and AI technologies for improved multi-omic data interpretation in drug discovery and clinical research, highlighting growing industry momentum. Additionally, recent U.S. FDA approvals of AI-enabled tools for liver disease clinical trials demonstrate increasing regulatory acceptance. Together, these advancements are accelerating biomarker discovery, improving prediction accuracy, and enabling faster, more cost-effective clinical trials ultimately advancing the development of precision medicine.
Growth Factors
- Rising Demand for Precision Medicine: Increasing focus on personalized therapies is driving the adoption of omics-based clinical trials to match treatments with patient-specific genetic and molecular profiles.
- Advancements in Sequencing and Multi-Omics Technologies: Continuous improvements in next-generation sequencing (NGS), proteomics, and integrated multi-omics platforms are enabling deeper biological insights and more efficient trial designs.
- Integration of AI and Advanced Bioinformatics: AI-powered analytics are accelerating biomarker discovery, patient stratification, and outcome prediction, making omics-based trials more accurate and cost-effective.
- Growing Pharmaceutical and Biotech R&D Investments: Drug developers are increasing spending on biomarker-driven and targeted therapies, boosting demand for omics-enabled trial methodologies.
- Expanding Collaboration Ecosystems: Strong partnerships among CROs, academic institutions, and technology providers are supporting large-scale data generation, interpretation, and trial execution.
Regional Insights
North America dominates the omics-based clinical trials market due to strong biotech and pharmaceutical presence, high R&D investments, advanced healthcare infrastructure, and early adoption of AI and precision medicine approaches. Europe follows closely, supported by robust genomics research programs, favorable regulatory frameworks, and increasing cross-border clinical research collaborations.
The Asia-Pacific region is expected to witness the fastest growth, driven by expanding clinical trial activity, growing genomics capabilities, government-backed precision medicine initiatives, and rising participation from China, Japan, South Korea, and India. Meanwhile, Latin America and the Middle East & Africa are emerging markets, gradually gaining traction as clinical research infrastructure improves and international sponsors expand trials into these regions for diverse patient populations and cost advantages.
Omics-Based Clinical Trials Market Key Players
- Parexel International (MA) Corporation
- Thermo Fisher Scientific Inc.
- Charles River Laboratories
- ICON plc
- SGS Sociwete Generale de Surveillance SA
- Eli Lilly and Company
- Pfizer Inc.
- Laboratory Corporation of America
- Novo Nordisk A/S
- Rebus Biosystems, Inc.
Recent Developments
- In April 2024, Signios Bio launched its AI-driven bioinformatics and advanced multiomics platform, rebranding to accelerate precision medicine and drug discovery with integrated multi-omic analyses for novel biomarkers and therapeutic insights.
- In January 2025, Enhanc3D Genomics introduced a suite of 3D multi-omics solutions using its proprietary platform to provide high-resolution, genome-wide data across drug discovery stages, enhancing target identification and disease mechanism insights.
- In December 2025, Valinor secured US$ 13 million in seed funding to deploy AI trained on matched multi-omic and clinical data to predict patient responses and reduce clinical trial failures, potentially speeding drug development and lowering R&D costs
Segments Covered in the Report
By Omics Type
- Genomics
- Transcriptomics
- Proteomics
- Metabolomics
- Epigenomics
- Microbiomics
- Multi-Omics (Integrated Platforms)
By Study Phase
- Phase I
- Phase II
- Phase III
- Phase IV
By Application
- Oncology
- Rare & Genetic Diseases
- Neurology
- Cardiovascular Diseases
- Immunology & Inflammatory Disorders
- Infectious Diseases
- Metabolic Disorders
By End-user
- Pharmaceutical Companies
- Biotechnology Companies
- Contract Research Organizations (CROs)
- Academic & Research Institutes
- Hospitals & Clinical Research Centers
By Service Type
- Trial Design & Biomarker Strategy
- Sample Collection & Biobanking
- Omics Data Generation
- Bioinformatics & Data Analytics
- Regulatory & Submission Support
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Get Sample Link : https://www.precedenceresearch.com/sample/7259
