The global battery free implants market size is valued to reach around USD 43 55 billion by 2034 increasing from USD 7 99 billion in 2024, with a CAGR of 18.48%.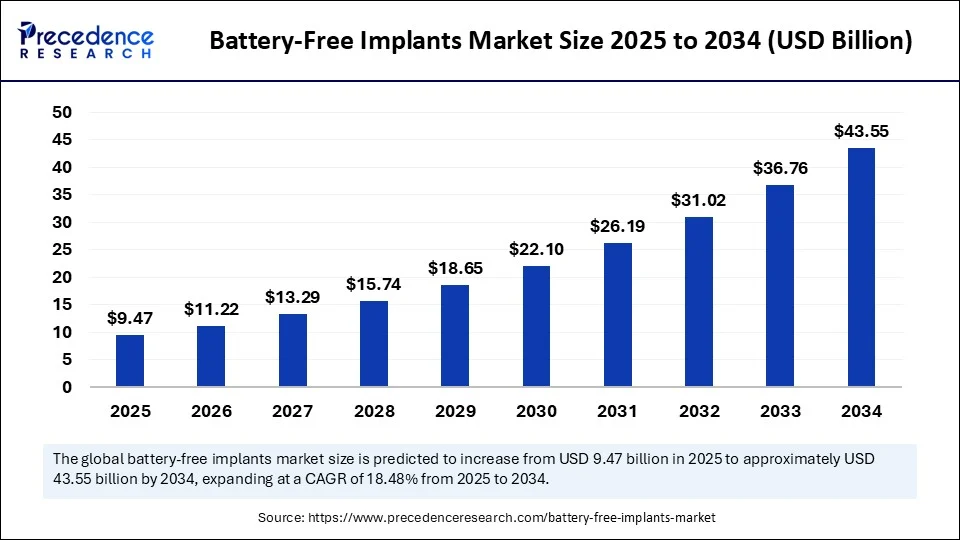
Battery Free Implants Market Key Takeaways
- In terms of revenue, the global battery-free implants market was valued at USD 7.99 billion in 2024.
- It is projected to reach USD 43.55 billion by 2034.
- The market is expected to grow at a CAGR of 18.48% from 2025 to 2034.
- North America dominated the global battery-free implants market in 2024.
- Asia Pacific is expected to grow at the fastest CAGR from 2025 to 2034.
- By application, the cardiac monitoring and pacing devices segment held the major market share in 2024.
- By application, the orthopedic monitoring devices segment is projected to grow at the highest CAGR between 2025 and 2034.
- By therapeutic area, the cardiology segment contributed the biggest market share in 2024.
- By therapeutic area, the orthopedics segment is expanding at a significant CAGR between 2025 and 2034.
- By energy harvesting technology, the radiofrequency (RF)-based devices segment led the market in 2024.
- By energy harvesting technology, the piezoelectric energy conversion segment is expected to grow at a significant CAGR over the projected period.
- By material, composite materials generated the largest market share in 2024.
- By material, bioresorbable materials segment is expected to grow at a notable CAGR from 2025 to 2034.
- By end user, the hospitals and clinics segment held the major market share in 2024.
- By end user, the homecare settings segment is projected to grow at a significant CAGR between 2025 and 2034
Impact of Artificial Intelligence on the Battery-Free Implants Market
Artificial intelligence is revolutionizing battery-free implant technology, enabling faster, smarter, and more precise clinical integration. It leverages AI-based modeling to optimize energy-harvesting systems—using body heat and movement—to ensure stable performance in challenging physiological conditions. In clinical environments, AI-driven data analytics identify the ideal candidates for these devices. Additionally, AI accelerates a shift toward more sustainable, minimally invasive, and customized implantable electronics by reducing reliance on batteries while enhancing device capabilities
Market Overview
The battery free implants market encompasses implantable medical devices that eliminate internal batteries by harvesting energy from the body or external sources. Relying on technologies like radiofrequency (RF), ultrasound, thermoelectric, piezoelectric, triboelectric generators or even biofuel cells, these implants deliver monitoring, neuromodulation, drug delivery or cardiac pacing without requiring battery replacements.
The elimination of internal power sources enables miniaturization, greater longevity, less invasive design and improved safety. Such self‑powered implants are rapidly gaining traction in applications where continuous, long‑term operation is critical, particularly for chronic disease management and remote patient monitoring.
Get Sample Link@ https://www.precedenceresearch.com/sample/6346
Market Drivers
-
Growth in chronic disease and aging demographics: Rising incidences of cardiovascular disease, diabetes, neurological conditions and an expanding elderly population are fueling demand for solutions that allow continuous monitoring or therapeutic intervention with minimal maintenance.
-
Shift toward minimally invasive care and home‑based treatment: Healthcare systems increasingly favor outpatient and home‑based models. Battery‑free implants reduce risks and logistical burden of surgeries for battery replacements, making them appealing for decentralized care.
-
Advances in energy‑harvesting and microelectronics: Innovations in RF wireless power transfer, piezoelectric/triboelectric nanogenerators, thermoelectric harvesters and biofuel cells have improved efficiency, power density, and miniaturization, making battery‑free devices more practical.
-
Regulatory momentum and innovation funding: Governments and regulatory bodies in the U.S., EU and Asia are prioritizing safe, less invasive implantable devices. Funding from national health institutes and regulatory fast‑track programs support commercialization.
-
Telehealth and AI integration: The expansion of remote care platforms encourages integration of implantable sensors that communicate wirelessly. AI-driven analytics enable adaptive therapies, predictive monitoring and personalization.
Opportunities
-
Personalized medicine & AI‑powered adaptive therapies: Battery‑free devices can collect physiological data continuously and respond in real‑time with tailored therapies. Neuromodulation implants could automatically adjust stimulation patterns based on the patient’s movement or symptoms.
-
Partnerships across healthcare and semiconductor ecosystems: Collaborations with chip makers, energy‑harvesting specialists and bioelectronics research labs accelerate innovation in ultra‑low‑power circuits, materials, and wireless interfaces.
-
New applications in under‑explored clinical areas: Emerging use cases include battery‑free implants in fertility monitoring, ophthalmologic sensors, orthopedic force measurement, endocrine sensing and responsive drug delivery systems.
-
Biodegradable, resorbable implants: Development of implants that dissolve after completing their function avoids secondary surgeries, opens applications in temporary diagnostic or therapeutic treatments, and appeals to eco‑conscious providers.
-
Emerging markets and aging societies: As middle‑income regions expand telemedicine and preventive care, battery‑free implants offer long‑term cost savings and accessibility in markets like India, China and Latin America.
Challenges
-
Energy reliability & deep‑tissue power constraints: Harvesting enough consistent power for critical implants like pacemakers or neurostimulators is technically challenging, especially when implants are deep within the body or misaligned relative to external power sources.
-
High development and manufacturing costs: Microfabrication of biocompatible, implantable systems and prolonged clinical testing lead to high R&D costs, which translate into expensive devices that may limit accessibility.
-
Limited long‑term clinical data and adoption lag: As a newer class of devices, battery‑free implants still lack extensive real‑world validation over years or decades. Physicians and payers may delay adoption until long‑term safety and efficacy data is available.
-
Regulatory and interoperability complexity: Diverse requirements across regions in safety, electromagnetic exposure (SAR), cybersecurity and clinical integration slow down global rollout.
-
Cybersecurity and connectivity risks: Wireless communication and integration with remote systems introduce vulnerabilities requiring robust encryption, authentication and safety protocols.
Recent Developments
-
A biotech startup pioneered a 4 mm diameter battery‑free pacemaker using additive manufactured electronics, capable of operating on nano‑watt power and deployable through minimally invasive catheter routes.
-
University labs and research institutions have demonstrated ultra‑small soft‑metal circuits and coils harvested up to 178 mW/cm² via inductive coupling, enabling practical energy yield for optogenetic and sensor implants.
-
Mechanical metamaterial implants capable of self‑harvested tactile sensing have been developed without electronics, opening possibilities for orthopedic force‑sensing implants.
-
New MXene‑based soft bioelectronic patches have shown low‑impedance interfacing and wireless stim/recording, combining battery‑free energy harvesting and tissue conformity.
-
Regulatory authorities in the U.S. FDA and EU MDR are advancing guidelines specific to wireless self‑powered implants, reducing barriers to approval for qualified devices.
-
Hybrid systems linking external wearable transmitters with internal sensors are gaining traction—enabling bidirectional communication, adaptive therapy control and enhanced battery‑free performance.
Battery-Free Implants Market Companies
- Abbott Laboratories
- Biotronik SE & Co. KG
- Cochlear Limited
- EBR Systems, Inc.
- Medtronic plc
- NeuroPace Inc.
- Pixium Vision
- Profusa Inc.
- Second Sight Medical Products Inc.
- Stimwave Technologies Inc.
Latest Announcement by Industry Leader
- In April 2025, CELTRO, established in late 2019, is a forward-thinking startup formed by heart rhythm specialists and experts from the semiconductor industry, including seasoned engineers and executives. The company strives to transform the integration of implants within the human body by designing electronic devices that function independently of external power sources. CELTRO’s inaugural initiative involves the development of a 3D-printed, battery-free pacemaker implant utilizing advanced additively manufactured electronics (AME) technology. “We are extremely excited by the progress we’ve made in the development and testing of our battery-free cardiac implant that has been enhanced using Nano Dimension’s AME technology. We are optimistic about the prospects of creating a platform of next-generation in-body electronics that will help shape the future of the medical implant industry” – Dr.-Ing Gerd Teepe, CELTRO Co-Founder & CEO.
Segments covered in the report
By Application
- Neural Stimulation Devices
- Cardiac Monitoring & Pacing Devices
- Drug Delivery Systems
- Bio-sensing and Diagnostics
- Hearing Implants
- Orthopedic Monitoring Device
By Therapeutic Area
- Cardiology
- Neurology
- Orthopedics
- Endocrinology (e.g., Glucose Monitoring)
- ENT (Ear, Nose, Throat)
- Urology & Gastroenterology
By Energy Harvesting Technology
- Radiofrequency (RF)-Based Devices
- Ultrasound Energy Harvesting
- Piezoelectric Energy Conversion
- Magnetic Resonance Coupling
- Thermoelectric & Bioelectric Harvesting
By Material Type
- Biocompatible Polymers
- Titanium & Other Metals
- Ceramic-Based Materials
- Bioresorbable Materials
- Composite Materials
By End User
- Hospitals & Clinics
- Ambulatory Surgical Centers
- Research & Academic Institutes
- Homecare Settings
- Specialty Clinics
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East
- Africa
Also Visit @https://www.precedenceresearch.com/
