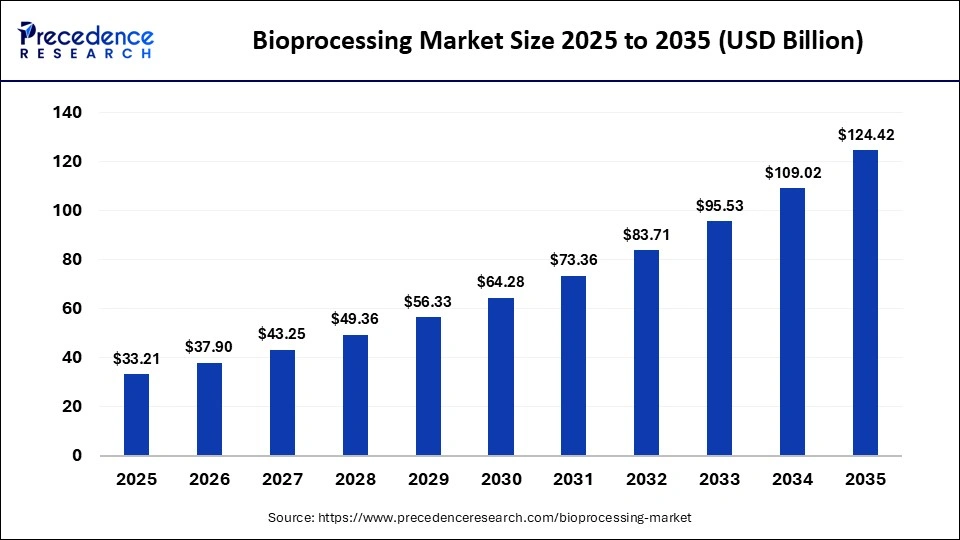
Market Key Takeaways
- North America led the bioprocessing market with a 46.7% market share in 2035.
- Asia Pacific is expected to expand at the fastest CAGR of 12.5% between 2026 and 2035.
- By product, the upstream bioprocessing products segment captured around 58.9% of the market share in 2025.
- By product, the single-use bioprocessing products segment is growing at a strong CAGR of 10.8% from 2026 to 2035.
- By type, the bioreactors segment contributed the highest market share of 34.8% share in 2025, with single-use bioreactors segment expected to expand fastest CAGR of 11% over the projected period 2026 and 2035.
- By application, the monoclonal antibody production segment captured 38.6% of market share in 2025.
- By application, the cell & gene therapy segment is expanding at the highest CAGR of 11.4% between 2026 and 2035.
- By capacity, the large-scale bioprocessing segment led with a 56.8% share in 2025.
- By capacity, the small-scale bioprocessing segment is set to grow at the highest CAGR of 11.3% between 2026 and 2035.
- By end-user, the pharmaceutical & biotechnology companies segment held more than 43.4% of the market share in 2024.
- By end-user, the contract development & manufacturing organizations (CDMOs) segment is expected to expand at the fastest CAGR of 11.5% between 2026 and 2035.
How Bioprocessing Is Transforming Healthcare Delivery
Bioprocessing uses living cells, enzymes, and biological systems to manufacture complex therapeutic products such as vaccines, biologics, cell and gene therapies, and recombinant proteins. These processes enable the large-scale, controlled production of advanced biological molecules that cannot be efficiently created through traditional chemical methods. As demand rises for biologics, cell and gene therapies, and precision medicine, bioprocessing has become one of the fastest-growing segments within the pharmaceutical and medical manufacturing landscape.
To improve efficiency and product quality while reducing contamination risks, bioprocessing companies are increasingly adopting continuous manufacturing, single-use systems, and advanced upstream production technologies. These approaches allow for greater flexibility, faster scale-up, and lower operational costs. In parallel, the integration of automation, digital twin technologies, and AI-driven process optimisation is improving batch-to-batch consistency, shortening development timelines, and enhancing overall process control. Ongoing investments in biologics and viral vector manufacturing capacity, along with expanding partnerships between biopharmaceutical companies and contract development and manufacturing organisations (CDMOs), are further strengthening the bioprocessing ecosystem and supporting its continued growth to meet global healthcare demands.
AI-Driven Innovation in the Modern Bioprocessing Market
Artificial intelligence has emerged as a key driver in the evolution of bioprocessing, reshaping how biologics are developed, monitored, and scaled. AI-powered technologies are increasingly being embedded across bioprocessing workflows to enhance efficiency, accuracy, and scalability. A notable example is the July 2025 announcement by a Canada UK research consortium, which launched an AI-integrated bioreactor as part of a coordinated AI–biotech experiment, highlighting the growing convergence of advanced analytics and biomanufacturing.
Machine learning tools now enable researchers and biopharmaceutical manufacturers to optimise cell line development, refine growth conditions, and predict cell behaviour using real-time process data. This shift supports a digital twin based production model, where virtual replicas of bioprocesses are used for simulation, predictive analytics, and autonomous upstream control. As AI technologies continue to advance, they are helping to reduce process variability, accelerate development timelines, and improve batch-to-batch consistency. Collectively, these innovations are transforming bioprocessing into a more precise, efficient, and data-driven manufacturing environment, well suited to the growing demands of modern biologics production.
Major Advances Shaping the Bioprocessing Market Today
- Single-Use Systems: The adoption of single-use bioreactors, filtration units, and disposable components is accelerating across the bioprocessing industry. These systems offer greater operational flexibility, significantly reduce contamination risks, and enable faster changeovers between production runs. A key advantage is their suitability for small-batch biologics production, which lowers capital investment, minimizes facility maintenance costs, and eliminates the need for complex cleaning and validation procedures.
- Continuous Bioprocessing: Continuous bioprocessing is transforming biologics manufacturing by enabling higher productivity with consistent product quality. This approach allows real-time process monitoring, improved yields, and a smaller manufacturing footprint compared to traditional batch processes. Continuous systems also support more efficient scale-up and are particularly valuable for producing complex biologics and advanced therapies with greater control and reproducibility.
- Growing Use of Automation and AI: The integration of automation and artificial intelligence is enhancing process visibility and operational efficiency across upstream and downstream bioprocessing stages. AI-driven analytics enable early detection of process deviations, predictive maintenance, and reduced reliance on manual intervention. These technologies improve reliability, optimize workflows, and ensure greater batch-to-batch consistency, ultimately leading to more robust and scalable production environments.
- Rising Demand for Cell and Gene Therapies: Increasing demand for viral vectors, personalized medicines, and next-generation therapeutics is driving innovation in scalable and closed-system bioprocessing solutions. Manufacturers are investing in advanced purification technologies, improved cell culture management, and flexible production platforms to support the complex requirements of cell and gene therapies, further reshaping the bioprocessing landscape.
Regional Insights: Bioprocessing Market
North America leads the global bioprocessing market, driven by a strong biopharmaceutical industry, advanced research infrastructure, and high adoption of innovative manufacturing technologies. The United States, in particular, dominates due to significant investments in biologics, vaccines, and cell and gene therapies, supported by robust funding from both government and private sectors. Early adoption of single-use systems, continuous bioprocessing, and AI-driven automation is strengthening manufacturing efficiency and scalability across the region.
Europe represents a mature and innovation-driven market, supported by strong regulatory frameworks, sustainability initiatives, and a well-established biopharmaceutical ecosystem. Countries such as Germany, the UK, Switzerland, and France are key contributors, with growing adoption of continuous processing and advanced automation. The region also benefits from increasing collaboration between biopharmaceutical companies and contract development and manufacturing organisations (CDMOs) to expand biologics and advanced therapy production capacity.
The Asia Pacific region is the fastest-growing bioprocessing market, fueled by expanding biopharmaceutical manufacturing, rising healthcare demand, and strong government support for biotechnology development. China, India, South Korea, Japan, and Singapore are emerging as major bioprocessing hubs, with investments in large-scale biologics production facilities, vaccine manufacturing, and cell and gene therapy capabilities. Cost-effective manufacturing and a growing CDMO presence further accelerate regional growth.
Top Players in the Bioprocessing Market and Their Offerings
- Thermo Fisher Scientific
- Sartorius AG
- Merck KGaA
- Danaher Corporation
- GE Healthcare
- Lonza Group
- Eppendorf AG
- Boehringer Ingelheim BioXcellence
- Corning Incorporated
- Repligen Corporation
- Fujifilm Diosynth Biotechnologies
- PBS Biotech
- Novasep
- Getinge AB
- Agilent Technologies
Recent Developments
- In December 2025, Ami Polymer launched IMAFLEXELL Film, a new 5-layer multi-layer barrier film for single-use bioprocessing systems, offering superior gas barrier, mechanical strength, chemical compatibility, and sterility for various biopharma applications.
- In January 2025, Repligen introduced its CTech SoloVPE PLUS System, a UV-Vis variable-pathlength analyser delivering accurate protein, DNA/RNA, or mRNA concentration measurements in under 30 seconds, streamlining bioprocess analytics workflows.
- In May 2025, Asahi Kasei Bioprocess America and PeptiSystems announced an exclusive global partnership to supply AKBA’s THESYS ACS Ergo synthesis columns to PeptiSystems’ peptide synthesis platforms, aiming to streamline and scale up peptide and oligonucleotide drug manufacturing.
- In June 2025, Ecolab Life Sciences launched Purolite AP+50, a new affinity chromatography resin with high dynamic binding capacity and durability, designed to improve cost-efficiency and throughput in large-scale monoclonal-antibody manufacturing.
Get Sample Link : https://www.precedenceresearch.com/sample/7224
