Blood Transfusion Diagnostics Market Key Takeaways
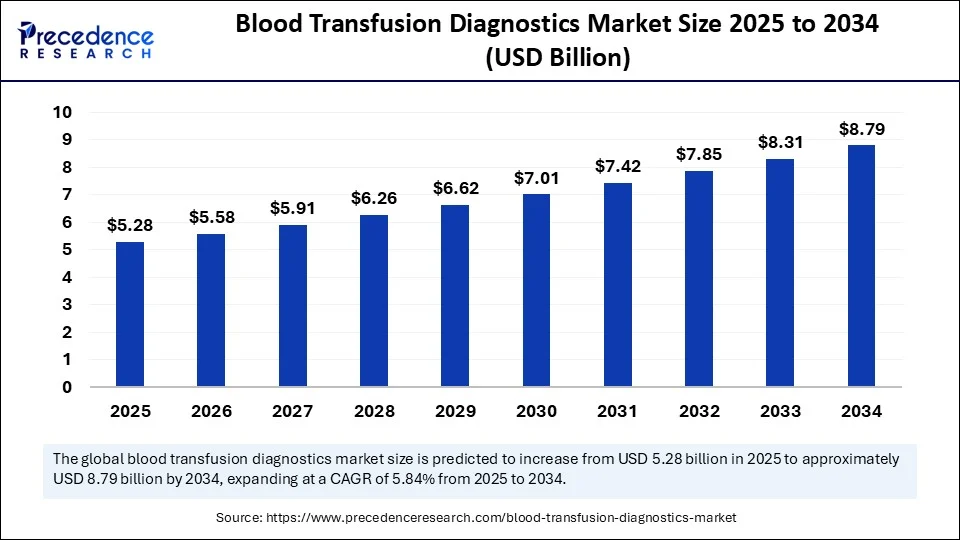
- In terms of revenue, the global blood transfusion diagnostics market was valued at USD 4.98 billion in 2024.
- It is projected to reach USD 8.79 billion by 2034.
- The market is expected to grow at a CAGR of 5.84% from 2025 to 2034.
- North America dominated the blood transfusion diagnostics market with the largest market share of 33.7% share in 2024.
- Asia Pacific is expected to expand at the fastest CAGR between 2025 and 2034.
- By product type, the reagents & kits segment held the biggest market share of 52.8% in 2024.
- By product type, the instruments segment is expected to grow at the fastest CAGR between 2025 and 2034.
- By test type, the disease screening segment captured the biggest market share of 45.1% in 2024.
- By test type, the molecular testing segment is expected to expand at a notable CAGR over the projected period.
- By technology, the serology-based testing segment contributed the major market share of 48.6% in 2024.
- By technology, the molecular-based testing segment is expected to expand at the highest CAGR over the projected period.
- By end user, the hospitals segment generated the major market share of 42% in 2024.
- By end user, the plasma fractionation centers segment is expected to expand at a notable CAGR over the projected period.
How AI Is Redefining Blood Transfusion Diagnostics
Artificial intelligence (AI) is transforming blood transfusion diagnostics by enhancing accuracy, efficiency, and patient safety. Through predictive modeling, decision support systems, and automated workflows, AI is streamlining laboratory processes, accelerating test results, and minimizing human error. It also enables early detection of potential complications, such as transfusion reactions, allowing clinicians to take timely, preventive measures.
In a groundbreaking development from January 2025, researchers at Emory University introduced an AI model trained on data from over 72,000 ICU patients. This model can predict transfusion needs within 24 hours, achieving an AUROC of 0.97 and an impressive 93% accuracy rate.
At DDW 2025, researchers from McGill University and Yale University unveiled a trajectory flow matching AI system capable of forecasting transfusion requirements and mortality risks in patients with acute gastrointestinal bleeding. This model demonstrated 93.6% accuracy, significantly outperforming conventional clinical tools.
Meanwhile, Canadian researchers at the University of British Columbia (UBC) are utilizing deep learning on microfluidic-sorted red blood cell (RBC) images to assess RBC deformability—an innovation that could improve storage protocols and matching strategies. Additionally, AIDA Diagnostics has developed a smart assistant that integrates neural networks with expert systems to recommend personalized transfusion volumes. This not only minimizes resource waste but also enhances clinical outcomes.
Market Overview
The blood transfusion diagnostics market comprises a wide array of products, instruments, and technologies designed to ensure the safety, compatibility, and effectiveness of blood and blood products used in transfusions. Key components include immunohematology testing, infectious disease screening, molecular diagnostics, and HLA typing. These diagnostic tools are critical for identifying blood group antigens, detecting transfusion-transmissible infections (TTIs), and confirming donor-recipient compatibility.
This market serves various end users, including blood banks, hospitals, transfusion centers, and diagnostic laboratories. As a cornerstone of modern transfusion medicine, blood transfusion diagnostics play a pivotal role in maintaining safe blood supplies, reducing the risk of adverse transfusion reactions, and supporting efficient clinical decision-making.
Blood Transfusion Diagnostics Market Growth Factors
- Increase in Surgical Procedures: The growth in complex surgeries, trauma care, and organ transplants is creating a need for pre-transfusion testing. This testing helps prevent adverse reactions and ensures patient safety during procedures.
- Advancements in Diagnostic Technologies: The use of molecular typing, nucleic acid testing, and automated platforms improves detection accuracy and processing speed. This progress enhances safety and efficiency in transfusion services.
- Stringent Safety Regulations: Regulatory bodies are enforcing strict blood screening protocols to prevent transfusion-transmitted infections (TTIs). This push is leading healthcare providers to adopt better diagnostic solutions worldwide.
Opportunity
Could Expanding Pathogen Reduction Systems Transform Blood Transfusion Diagnostics?
A major opportunity for the blood transfusion diagnostics market lies in scaling pathogen reduction technologies (PRTs), especially riboflavin and UV light systems, to eliminate transfusion-transmitted infections in donated blood components. A global health review shows that PRT significantly reduces risks from HIV, HBV, HCV, Zika, dengue, and malaria, even during emerging outbreaks. In 2024, the FDA-cleared Intercept Blood System for plasma uses amotosalen and UV light to inactivate viruses like West Nile, hepatitis viruses, and HIV.
In low- and middle-income countries, combining PRT with rapid diagnostics could greatly reduce transmission events. One modeling study estimated complete elimination for HIV and significant drops for HBV and HCV. As governments and blood services worldwide focus on safer transfusion practices, broad adoption of PRT provides diagnostic manufacturers with a strong growth path. This approach improves blood safety, increases compliance, and addresses important screening gaps.
Recent Developments
- In February 2025, HC1 and Accumen launched next-generation Clinical Excellence Solutions to optimize blood use and diagnostics. The platform uses AI and lab insights to improve transfusion decision-making, enhance patient outcomes, and help hospitals reduce unnecessary blood usage.
- In October 2024 Terumo Blood and Cell Technologies began the U.S. rollout of Reveos, an automated blood processing system. It allows for efficient separation of blood components, cutting down on manual steps and improving diagnostic accuracy for transfusion services in clinical laboratories and blood centers.
Blood Transfusion Diagnostics Market Companies

- Grifols, S.A.
- Ortho Clinical Diagnostics
- Bio-Rad Laboratories, Inc.
- F. Hoffmann-La Roche Ltd.
- Abbott Laboratories
- Siemens Healthineers AG
- Immucor, Inc.
- Thermo Fisher Scientific Inc.
- Quotient Limited
- Danaher Corporation (Beckman Coulter)
- Becton, Dickinson and Company (BD)
- HemoCue AB
- BioMérieux SA
- DiaSorin S.p.A.
- Sysmex Corporation
- Bag Health Care GmbH
- Fresenius Kabi AG
- Gen-Probe Incorporated (Hologic)
- Agena Bioscience, Inc.
- Tulip Diagnostics (P) Ltd.
Recent Developments
- In February 2025, HC1 and Accumen launched next-generation Clinical Excellence Solutions to optimize blood use and diagnostics. The platform uses AI and lab insights to improve transfusion decision-making, enhance patient outcomes, and help hospitals reduce unnecessary blood usage.
- In October 2024 Terumo Blood and Cell Technologies began the U.S. rollout of Reveos, an automated blood processing system. It allows for efficient separation of blood components, cutting down on manual steps and improving diagnostic accuracy for transfusion services in clinical laboratories and blood centers.
Segments Covered in the Report
By Product Type
- Instruments
- Immunohematology Analyzers
- Nucleic Acid Amplification Systems (NAT)
- Microarray Platforms
- ELISA Readers
- PCR Systems
- Others
- Reagents & Kits
- ABO/Rh Typing Reagents
- Antibody Screening Reagents
- NAT Reagents
- HLA Typing Kits
- ELISA Kits
- Others
- Software & Services
- Blood Bank Management Software
- Laboratory Information Systems (LIS)
- Testing Services
- Others
By Test Type
- Blood Group Typing
- ABO Typing
- Rh Typing
- Antibody Screening
- Disease Screening
- HIV
- Hepatitis B & C
- Syphilis
- Malaria
- Other
- Molecular Testing
- NAT (Nucleic Acid Testing)
- Genotyping & HLA Typing
- Pathogen Inactivation Detection
- Crossmatching & Compatibility Testing
- Major Crossmatch
- Minor Crossmatch
By Technology
- Serology-based Testing
- Molecular-based Testing
- Microarray
- Next-generation Sequencing (NGS)
- Enzyme-linked Immunosorbent Assay (ELISA)
- Rapid Diagnostics
- Others
By End User
- Hospitals
- Blood Banks
- Diagnostic Laboratories
- Plasma Fractionation Centers
- Academic & Research Institutes
Others
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
