Key Takeaways
- North America led the market in 2025 with a share of around 40%.
- Asia Pacific is expected to grow at a CAGR of 6.5% during the forecast period.
- By product type, the dexamethasone segment held the largest market share of 35% in 2025.
- By product type, the Tobradex segment is expected to expand at a CAGR of 5.1% during the forecast period.
- By application, the adult segment held the largest market share of 80% in 2025.
- By application, the children segment is expected to grow at a solid CAGR of 5.3% between 2026 and 2035.
- By distribution channel, the hospital pharmacy segment held the highest market share of 45% in 2025.
- By distribution channel, the online pharmacy segment is expected to grow at the fastest CAGR of 5.7% during the forecast period.
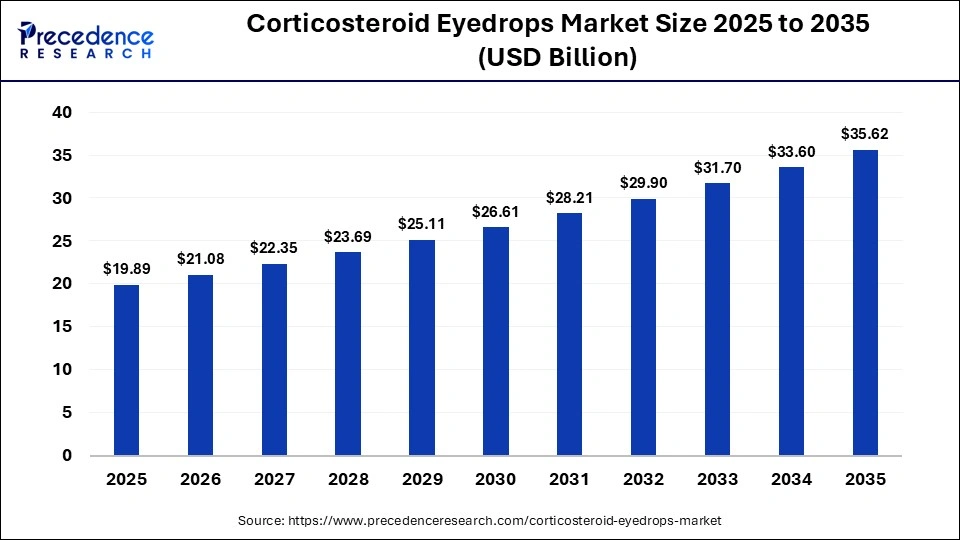
Market Overview
The corticosteroid eyedrops market includes ophthalmic steroid formulations used to manage ocular inflammation, post-operative swelling, uveitis, allergic reactions, and other inflammatory eye disorders. These medications function by suppressing immune responses in ocular tissues, thereby reducing redness, swelling, pain, and irritation.
Market growth is primarily driven by the rising global prevalence of eye diseases, increasing volume of ophthalmic surgeries, and expanding geriatric populations that are more susceptible to inflammatory and degenerative eye conditions. The rapid expansion of healthcare infrastructure, improved access to ophthalmic care, and the growing presence of online pharmacy distribution channels are further contributing to market expansion.
Impact of Artificial Intelligence on the Corticosteroid Eyedrops Market
Artificial Intelligence (AI) is transforming the corticosteroid eyedrops market by enabling a shift toward more precise, data-driven, and personalized ophthalmic care. AI technologies are accelerating drug discovery and formulation optimization by analyzing vast datasets related to chemical structures, pharmacokinetics, and physicochemical properties. This helps improve solubility, stability, and bioavailability of corticosteroid formulations while minimizing adverse effects.
AI is also enhancing therapeutic adherence through smart monitoring tools and predictive analytics that support personalized dosing strategies. In drug development, AI-powered platforms can identify promising molecular combinations faster, reducing research timelines and improving clinical success rates.
In January 2026, Insitro acquired CombinAbleAI to strengthen its AI-driven drug discovery capabilities. The acquisition supports the launch of an advanced AI-based platform named TherML, aimed at accelerating therapeutic innovation across multiple treatment areas, including ophthalmology.
Corticosteroid Eyedrops Market Trends
- Opening of New Production Centers: Several ophthalmic manufacturers and hospital-linked pharmaceutical units are expanding production capacities to meet growing demand. In October 2025, Aurolab inaugurated a new production facility in Madurai to manufacture approximately 36 million bottles of eyedrops annually, strengthening domestic supply and export capabilities.
- Growing Incidence of Eye Diseases: The increasing global burden of eye disorders such as uveitis, glaucoma, allergic conjunctivitis, and post-surgical inflammation is significantly boosting demand for corticosteroid eyedrops. According to projections by the American Academy of Ophthalmology, nearly 7.32 million individuals in the United States are expected to develop primary open-angle glaucoma by 2050, highlighting the rising need for effective ophthalmic treatments.
- Regulatory Approvals Supporting Market Expansion: Approvals from health authorities are expanding product availability and strengthening market penetration. In December 2024, the U.S. Food and Drug Administration approved Omnipred, an adrenocortical steroid-based eyedrop indicated for inflammatory ocular conditions such as acne rosacea and superficial punctate keratitis.
- Rising Adoption of Advanced Formulations: There is growing preference for preservative-free and sustained-release corticosteroid eyedrop formulations designed to improve patient tolerability and compliance. These advanced formulations aim to reduce dosing frequency and minimize side effects commonly associated with conventional steroid therapies.
- Growth of Combination Therapies: Pharmaceutical companies are increasingly launching combination ophthalmic products that integrate corticosteroids with antibiotics or antifungal agents. These dual-action formulations provide broader therapeutic coverage by addressing inflammation and potential infections simultaneously, enhancing treatment efficiency and convenience for patients.
Regional Insights
North America holds a significant share of the corticosteroid eyedrops market, driven by advanced healthcare infrastructure, high awareness of eye health, and strong adoption of innovative ophthalmic therapies. The United States leads the region due to rising cases of glaucoma, uveitis, allergic conjunctivitis, and post-surgical inflammation. The presence of established pharmaceutical companies, favorable reimbursement policies, and frequent regulatory approvals further support market growth. Additionally, the increasing volume of cataract and refractive surgeries contributes to sustained demand for anti-inflammatory ophthalmic medications.
Europe represents a mature and stable market, supported by well-developed public healthcare systems and strong regulatory oversight. Countries such as Germany, France, the United Kingdom, Italy, and Spain demonstrate consistent demand due to aging populations and high ophthalmic surgery rates. Growing adoption of preservative-free and sustained-release formulations aligns with the region’s emphasis on patient safety and quality standards. Furthermore, research collaborations and pharmaceutical innovation in ophthalmology continue to strengthen the regional market landscape.
Asia Pacific is projected to register the fastest growth in the corticosteroid eyedrops market. Rapid urbanization, increasing healthcare expenditure, and expanding access to ophthalmic services are key growth drivers. Countries such as China, India, Japan, and South Korea are witnessing rising incidences of eye disorders due to aging populations, diabetes prevalence, and increased screen exposure. Expanding local manufacturing capabilities and government initiatives aimed at improving eye care infrastructure are also accelerating market penetration. The growth of online pharmacies and retail drug chains further enhances product accessibility in the region.
Corticosteroid Eyedrops Market Key players
- Pfizer Inc.
- AbbVie Inc.
- Novartis AG
- Alcon Inc.
- Bausch Health Companies Inc.
- Sun Pharmaceutical Industries Ltd.
- Cipla Ltd.
- Santen Pharmaceutical Co. Ltd.
- Lupin Limited
- Zydus Lifesciences Ltd.
- Glenmark Pharmaceuticals Ltd.
- Alkem Laboratories Ltd.
- Sunways Pvt. Ltd.
- FDC Limited
- Akorn Pharmaceuticals Inc.
Recent Developments
- In February 2026, Formosa Pharmac collaborated with Arrotex Pharmaceuticals. This collaboration is aimed at launching clobetasol propionate ophthalmic suspension in New Zealand and Australia.
- In July 2025, Lupin launched Lotemax generic suspension. This suspension contains a corticosteroid effective for treating mycobacterial infection of the human eye.
- In April 2025, Nordic Pharma Inc partnered with Harrow. Through this partnership, these companies launched a corticosteroid eyedrop named ‘Maxitrol’ in the U.S.
Segments Covered in the Report
By Product Type
- Dexamethasone
- Cortisone
- Tobradex
- Others
By Application
- Adult
- Children
By Distribution Channel
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
- Other Channels
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
Get Sample Link : https://www.precedenceresearch.com/sample/7935
