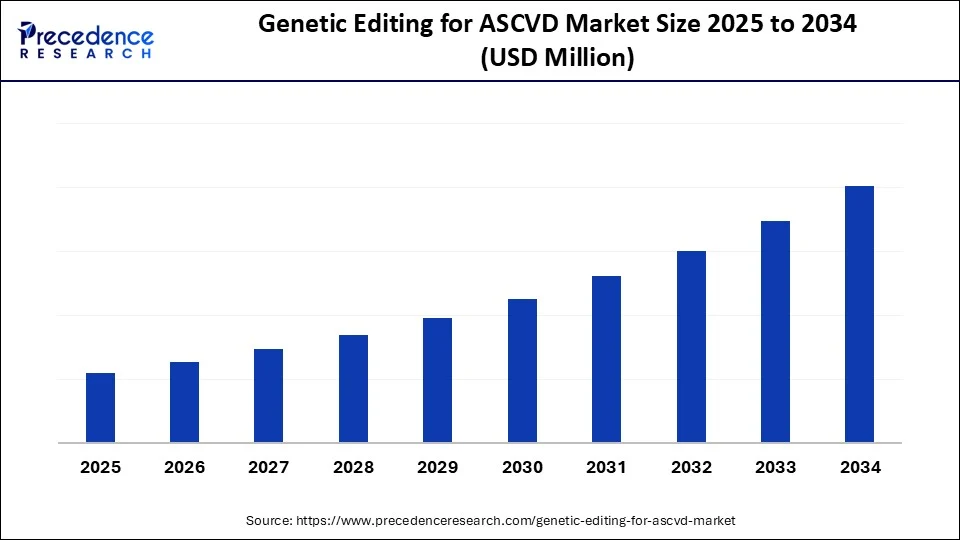
Genetic Editing for ASCVD Market Key Takeaways
- North America dominated the genetic editing for ASCVD market with the largest market share 52% in 2024.
- Asia Pacific is anticipated to witness the fastest growth during the forecast years.
- By editing modality, the CRISPR/Cas9 gene knockout segment captured the biggest market share of 44% in 2024.
- By editing modality, the base editing segment is anticipated to show considerable growth over the forecast period.
- By therapeutic target, the PCSK9 segment contributed the highest market share of 38% in 2024.
- By therapeutic target, the ANGPTL3 segment is anticipated to show considerable growth over the forecast period.
- By delivery system, the AAV vectors segment held a significant market share in 2024.
- By delivery system, the lipid nanoparticles segment is anticipated to show considerable growth over the forecast period.
- By stage of development, the preclinical research segment held a significant market share in 2024.
- By stage of development, the phase I/II clinical trials segment is anticipated to show considerable growth over the forecast period.
- By end user, the pharmaceutical and biotechnology companies segment generated the major market share in 2024.
- By end-user, the specialty cardiology clinics segment is anticipated to exhibit the fastest growth over the forecast period.
How Is AI Integration Transforming Genetic Editing for ASCVD?
Artificial Intelligence is playing a pivotal role in advancing genetic editing for atherosclerotic cardiovascular disease (ASCVD), significantly improving the speed, precision, and safety of gene therapy development. AI algorithms are capable of processing massive genomic datasets to identify ideal gene targets—such as PCSK9 or ANGPTL3—that are known to influence cholesterol regulation and cardiovascular risk.
By predicting off-target effects and refining target specificity, AI enhances the design and delivery accuracy of CRISPR-based therapies. This reduces the likelihood of unintended genetic modifications, thereby improving treatment safety and efficacy.
Beyond target identification, AI is accelerating the drug discovery process, streamlining experimental workflows, and enabling real-time monitoring of gene editing outcomes. Automation through AI also supports lab-scale optimization, from guide RNA selection to delivery method design, helping researchers iterate faster and bring therapies closer to clinical application.
As a result, AI integration is not only advancing the therapeutic potential of gene editing for ASCVD but also reshaping the entire development pipeline from discovery to clinical readiness.
Market Overview
The genetic editing for atherosclerotic cardiovascular disease (ASCVD) market refers to the application of next-generation gene editing technologies, such as CRISPR, base editing, and siRNA/ASO gene silencing, to prevent, treat, or reverse cardiovascular disorders caused by genetic dyslipidemia and inflammation, primarily familial hypercholesterolemia (FH), elevated Lp(a), PCSK9/LDLR mutations, and chronic vascular inflammation. The goal is to offer one-time or long-term disease-modifying treatments rather than lifelong lipid-lowering or anti-inflammatory drugs.
The genetic editing for ASCVD market is experiencing substantial growth, driven by advancements in CRISPR and gene therapy technologies. The rising prevalence of ASCVD, coupled with a growing demand for personalized medicine, is accelerating gene-targeted research. Increased funding for genomic research from government and private organizations, along with collaborations between biotech companies and healthcare networks, are further enhancing market growth. Moreover, the identification of genetic mutations, such as PCSK9 and ANGPTL3, linked to ASCVD, has opened up new avenues for therapeutic intervention.
What Factors are Fueling the Growth of the Genetic Editing for ASCVD Market?
- Innovations in Technology of Gene Editing: The discovery of gene editing tools such as CRISPR-Cas9, TALENs, and base editing has made gene editing easier, accurate, and less expensive. These technologies make it significantly easier to alter the genes implicated in ASCVD, including PCSK9 and ANGPTL3.
- Increment in the Frequency of ASCVD: Atherosclerotic cardiovascular disease continues to be a cause of mortality and morbidity in the world. The rising burden of the disease leads to an immediate need for new and long-term treatment methods, such as gene editing, where investments and research speed are accelerated.
- Long-term Industry Partnerships and Investment: The drug companies, start-up companies in the biotechnological field, and the university research centers are forging strategic partnerships to speed up the implementation of gene treatments for ASCVD. Such alliances increase research and development, as well as streamline clinical testing.
Opportunity
Advancement of Next-Generation Gene Editing Technologies
The emergence of advanced gene editing platforms and delivery systems is creating new opportunities to transform the treatment landscape for atherosclerotic cardiovascular disease (ASCVD). Innovations such as base editing, prime editing, and epigenome editing are expanding the possibilities beyond the limitations of traditional CRISPR-Cas9 systems.
These next-generation tools offer greater precision and reduced off-target activity, with the potential to repair genes without introducing double-stranded breaks. This not only improves the safety profile of gene therapies but also increases their applicability across a broader range of genetic targets.
As research continues to progress, these technologies are enabling the correction of multiple genetic risk factors simultaneously or the modulation of gene expression in adult patients. Such advancements could significantly enhance therapeutic outcomes and offer more effective, personalized treatment options for individuals at risk of ASCVD.
Regional Insights
North America maintained a leading position in the genetic editing for ASCVD market in 2024, supported by a well-developed infrastructure for biomedical innovation and genetic therapies. A combination of strong investment from venture capital firms, government agencies, and private companies continues to advance preclinical and clinical development in the region. Widespread awareness of personalized medicine, along with improved access to genetic testing, further contributes to market momentum.
The United States plays a central role in this growth. Factors such as an aging population, rising incidence of obesity and lipid disorders, and increasing healthcare expenditure have prompted greater interest in gene editing technologies. Regulatory support from agencies like the FDA, including expedited approvals and detailed guidance on genome-editing applications, has facilitated clinical progress. The country also hosts a significant number of early-phase clinical trials focused on cardiovascular gene editing, reinforcing its position as a leader in therapeutic innovation.
Asia Pacific is projected to grow at the fastest pace over the forecast period. This expansion is driven by rising investments in biotechnology, growing awareness of genetic conditions, and ongoing improvements in healthcare infrastructure. Countries including Japan, South Korea, India, and Singapore are actively shaping regulatory policies to support gene editing and precision medicine.
In particular, Japan has implemented accelerated regulatory pathways for genetic and regenerative treatments, which has encouraged the development of novel cardiovascular therapies. Increased affordability of genetic screening and the emergence of cross-sector partnerships are also contributing to growth in the region.
China is becoming an influential player in the space. With several gene editing projects entering early-stage clinical trials, China’s biotechnology ecosystem is showing strong interest in CRISPR-based approaches to treat cardiovascular and metabolic conditions. The country benefits from a growing genome data pool and a large base of skilled professionals in biomedical science. Although regulatory and ethical frameworks are still evolving, efforts are underway to align with international standards and strengthen oversight.
Europe is expected to see steady growth, supported by continued public and private investment in biomedical research, a comprehensive healthcare system, and the presence of prominent pharmaceutical companies and academic research centers. Research into gene editing therapies targeting ASCVD-related genes is actively ongoing, driven by the region’s need to address age-related cardiovascular health challenges.
The United Kingdom serves as a key contributor within the European landscape. Its well-established life sciences sector, academic excellence, and infrastructure for genomic research make it a hub for cardiovascular innovation. Institutions such as the UK Biobank, the Wellcome Sanger Institute, and the Francis Crick Institute play a vital role in collecting and analyzing genomic data. UK-based companies and research groups are developing gene editing solutions that target lipid metabolism and inflammatory pathways linked to ASCVD. Supportive regulatory mechanisms from national health agencies are helping accelerate clinical adoption of these advanced therapies.
Recent Developments
- In June 2025, Eli Lilly disclosed that it will acquire Verve Therapeutics in a transaction valued at around $1.3bn, representing a major augmentation to cardiovascular disease (CVD) capacity at the firm. The transaction will provide Lilly with access to a pipeline of single-course gene editing therapies that Verve is developing to treat the causes of atherosclerotic CVD (ASCVD), which contributes to approximately 85 percent of all cardiovascular fatalities and is brought on by the formation and proliferation of plaques in the innermost layer of the arteries.
- In May 2025, Ionis Pharmaceuticals, Inc. announced positive topline data of olezarsen in the Essence study of individuals with moderate hypertriglyceridemia (fasting triglycerides 150 mg/dL to less than 500 mg/dL) and with atherosclerotic cardiovascular disease (ASCVD) or at risk of the disease. Almost all the participants were using the present standard of care lipid-lowering medications.
- In April 2025, Verve Therapeutics, a clinical-stage company building a new class of genetic medicines to treat cardiovascular disease, announced positive initial data from the Heart-2 Phase 1b clinical trial of VERVE-102. The Heart-2 Phase 1b Investigation is measuring patients having heterozygous familial hypercholesterolemia (HeFH) and/or premature coronary artery disease (CAD), who necessitate profound and sustainable low-density lipoprotein cholesterol (LDL-C) levels in the blood.
Genetic Editing for ASCVD Market Top Companies

- CRISPR Therapeutics
- Intellia Therapeutics
- Editas Medicine
- Beam Therapeutics
- Verve Therapeutics
- Sangamo Therapeutics
- Vertex Pharmaceuticals
- Novartis
- Allogene Therapeutics
- Crispr BioPharma
- Alnylam Pharmaceuticals
- Arrowhead Pharmaceuticals
- Precision BioSciences
- BlueRock Therapeutics (Bayer)
- Acuitas Therapeutics
- Moderna Therapeutics
- Pfizer
- Amgen
- BioNTech
- Evotec
