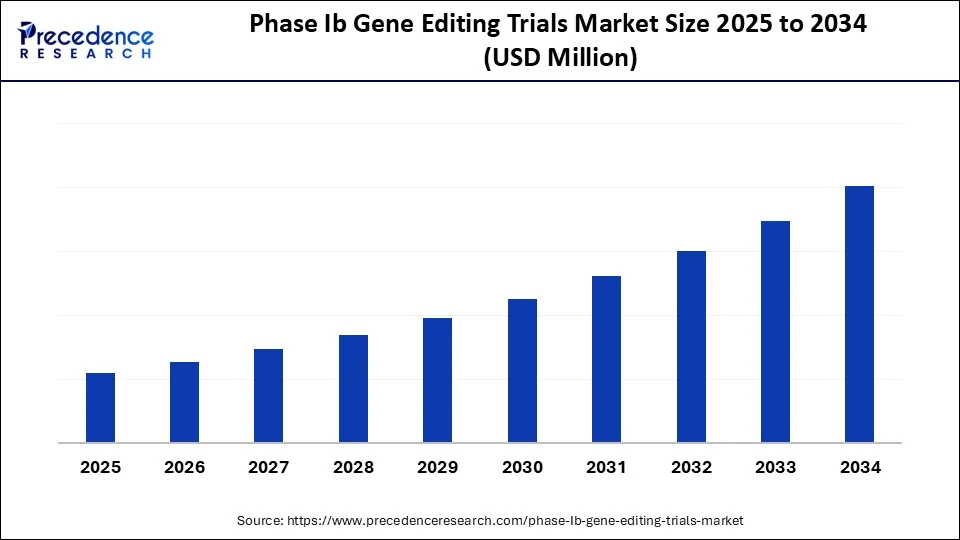
Phase Ib Gene Editing Trials Market Key Takeaways
- North America dominated the global phase Ib gene editing trials market with the largest share of 61% in 2024.
- Asia Pacific is expected to grow at the fastest CAGR from 2025 to 2034.
- By gene editing platform, the CRISPR/Cas systems segment held the biggest market share of 62% in 2024.
- By gene editing platform, the base and prime editors segment is projected to grow at a significant CAGR between 2025 and 2034.
- By therapy type, the in vivo gene editing therapies segment captured the biggest market share of 55% in 2024.
- By therapy type, the ex vivo gene editing therapies segment is likely to expand at a significant CAGR between 2025 and 2034.
- By disease area, the oncology segment contributed the highest market share of 38% in 2024.
- By disease area, the neurology segment is expected to grow at a significant CAGR over the projected period.
- By delivery method, the electroporation segment led the market in 2024.
- By delivery method, the non-viral vectors segment is expected to grow at a notable CAGR from 2025 to 2034.
- By trial sponsor type, the biotech/pharma companies segment contributed the highest market share of 73% in 2024.
- By trial sponsor type, the academic institutions segment is expected to grow at a notable CAGR from 2025 to 2034.
The Impact of Artificial Intelligence on the Phase Ib Gene Editing Trials Market
Artificial intelligence (AI) is playing a transformative role in Phase Ib gene editing trials, streamlining complex processes and enabling more informed, data-driven decisions. By leveraging advanced algorithms, AI can efficiently analyze vast datasets from preclinical and early-phase clinical studies, predict dose-response relationships, and identify potential off-target effects. This capability not only enhances the precision of clinical trial design but also supports real-time protocol optimization, significantly improving trial outcomes and success rates.
Market Overview
The Phase Ib gene editing trials market focuses on the early clinical evaluation of gene editing-based therapies—such as CRISPR, TALENs, and zinc finger nucleases (ZFNs)—in patients with target diseases. Phase Ib, a subsegment of Phase I, typically emphasizes proof-of-concept, preliminary efficacy signals, dose optimization, and extended safety assessment. Unlike Phase Ia trials, which often involve healthy volunteers, Phase Ib studies are generally conducted in small patient populations to determine the biological impact of the therapy in real-world disease contexts.
A key driver of this market is the rising prevalence of genetic disorders, prompting healthcare systems worldwide to prioritize early and targeted intervention strategies. Advanced gene editing tools—including CRISPR-Cas systems, base editing, and prime editing—allow scientists to correct faulty DNA sequences with unprecedented precision, fueling the development of novel therapeutics.
According to the Gene, Cell, & RNA Therapy Landscape Report, there are currently over 4,000 gene, cell, and RNA-based therapies in development globally. This surge in innovation is further supported by increasing venture capital investment in genome editing startups and the growing demand for precision medicine. Together, these trends are catalyzing an expansion of early-stage clinical trials, especially within the Phase Ib segment, across a wide range of therapeutic areas.
Phase Ib Gene Editing Trials Market Growth Factors
- Rising Demand for Rare Disease Therapies: Growing unmet clinical needs in treating rare monogenic disorders are fuelling the development of early-stage gene editing programs.
- Boosting Integration of AI in Trial Design: Integration of artificial intelligence tools is accelerating trial optimization, data analysis, and patient matching in Phase Ib studies.
- Expanding CRISPR Toolkits Beyond Cas9: The development of novel systems, such as Cas12, Cas13, and Cas14, is driving more versatile gene editing approaches for Phase Ib pipelines.
- Growing Academic: Industry Collaborations: Strategic alliances between research institutions and biotech firms are propelling translational research into early human trials.
Opportunity: How Growing Investments by Biotech Firms Accelerate the Phase Ib Gene Editing Pipeline
Rising investments from biotech firms are acting as a powerful catalyst for the expansion of the Phase Ib gene editing trial pipeline. Venture capital funding, strategic alliances with pharmaceutical companies, and public-sector grants are increasingly being directed toward enterprises developing next-generation gene editing technologies. These financial infusions are directly supporting the advancement of therapeutic candidates into early-phase clinical trials, particularly within the Phase Ib segment.
Leading companies such as CRISPR Therapeutics, Editas Medicine, and Beam Therapeutics are similarly channeling funds toward Phase Ib research initiatives to evaluate genome modification strategies in real-world disease models. These investments are vital in enabling high-complexity R&D programs, fostering innovation, and increasing the volume and quality of clinical trials.
Moreover, ongoing advancements in CRISPR, base editing, and prime editing technologies are further fueling early-phase clinical activity. As these tools evolve in precision and efficiency, they not only reduce development timelines but also improve the likelihood of clinical success—making the Phase Ib gene editing pipeline an increasingly attractive area for biotech investment.
Regional Insights
North America has become a leading region in the Phase Ib gene editing trials market due to its strong focus on advanced research infrastructure, a supportive regulatory environment, and consistent funding for biotechnology innovation. Several prominent biotech companies have initiated early-phase clinical trials across various therapeutic areas, reflecting the region’s commitment to progressing gene editing technologies.
Government agencies continue to play a major role in supporting this field. Key institutions are actively funding genome engineering initiatives, while regulatory authorities have streamlined the processes needed to begin and manage clinical trials. Collaborative efforts between academic institutions and industry partners have also contributed to this progress, enabling the transition of research into clinical applications.
Additional factors contributing to North America’s leadership include the presence of dedicated gene therapy centers, efficient patient recruitment processes, and access to real-time regulatory feedback. The growing investment in biomedical research and innovation further reinforces the region’s strong position in the early-stage development of gene editing therapies.
In comparison, the Asia Pacific region is showing significant momentum, driven by increased investment in biomedical research, improvements in regulatory frameworks, and rising collaboration between academia and industry. Countries across the region are supporting clinical development through national programs and funding schemes, which are accelerating the launch of early-phase gene editing trials. These advancements are expected to strengthen the region’s role in the market moving forward.
Recent Developments
- In June 2025, SNIPR Biome announced the initiation of its Phase 1b trial for SNIPR001, building on encouraging results from its Phase 1a trial in healthy volunteers. The earlier study, funded by CARB-X, demonstrated a favorable safety profile and effective target engagement. “This milestone builds upon the highly encouraging data from our Phase 1a trial, where SNIPR001 demonstrated promising safety and target engagement,” said Dr. Christian Grøndahl, Co-founder and CEO of SNIPR Biome.
- In January 2025, AskBio, a gene therapy firm under Bayer AG, announced the randomization of the first participants in its Phase II REGENERATE-PD clinical trial targeting Parkinson’s disease. Dr. Rajesh Pahwa, Director of the Parkinson’s Disease and Movement Disorder Center at the University of Kansas Medical Center and Principal Investigator of the study, noted, “The advancement of this important investigational gene therapy gives hope to patients and the medical community, especially given the critical need for neurorestorative therapies in Parkinson’s.”
- In May 2025, The Danforth Technology Company (DTC) unveiled the launch of Spearhead Bio, a startup dedicated to advancing plant genome engineering. The company’s proprietary Transposase Assisted Homology Independent Targeted Insertion (TAHITI) technology enables efficient and seamless gene integration in both transgenic and non-transgenic crops, aiming to enhance precision and efficiency in agricultural biotechnology.
Phase Ib Gene Editing Trials Market Top Companies

- Allogene Therapeutics
- Beam Therapeutics
- Caribou Biosciences
- Cellectis
- CRISPR Therapeutics
- EdiGene
- Editas Medicine
- Excision BioTherapeutics
- Graphite Bio
- iECURE
- Intellia Therapeutics
- LogicBio Therapeutics
- Metagenomi
- Poseida Therapeutics
- Precision BioSciences
- Regenxbio
- Sana Biotechnology
- Sangamo Therapeutics
- Tessera Therapeutics
- Verve Therapeutics
Also Read: AI in Clinical Trials Market
