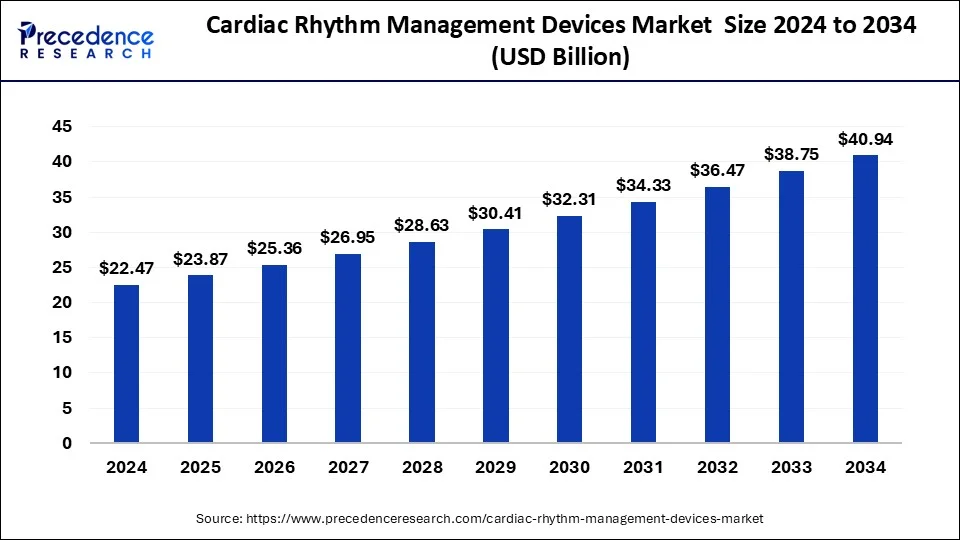
The global cardiac rhythm management devices market size reached USD 21.15 billion in 2023 and is projected to hit around USD 38.75 billion by 2033, poised to grow at a CAGR of 6.24% from 2024 to 2033.
Key Points
- North America has accounted more than 40% of the market share in 2023.
- By product, the defibrillators segment held the largest share of the market in 2023.
- By application, the arrhythmias segment held the largest share of the market in 2023.
- By end-user, the hospitals segment led the market with the largest market share of 50% in 2023.
The Cardiac Rhythm Management (CRM) Devices Market encompasses a range of medical devices designed to monitor and manage heart rhythm disorders, including arrhythmias and heart failure. These devices play a crucial role in diagnosing, treating, and preventing cardiac arrhythmias, which can lead to serious complications such as stroke, heart attack, or sudden cardiac death. The market for CRM devices has experienced steady growth due to increasing prevalence of cardiovascular diseases, advancements in technology, and rising demand for minimally invasive treatment options.
Get a Sample: https://www.precedenceresearch.com/sample/3968
Growth Factors:
Several factors contribute to the growth of the CRM Devices Market. Firstly, the rising prevalence of cardiovascular diseases, including atrial fibrillation, bradycardia, and heart failure, has driven the demand for CRM devices. As the aging population increases and lifestyles become more sedentary, the incidence of cardiac arrhythmias is expected to rise, fueling the demand for diagnostic and therapeutic solutions.
Moreover, technological advancements in CRM devices have led to the development of more sophisticated and effective treatment options. Innovations such as implantable cardioverter-defibrillators (ICDs), cardiac resynchronization therapy (CRT) devices, and leadless pacemakers have improved patient outcomes by providing targeted therapy with fewer complications and better device longevity.
Additionally, growing awareness about the benefits of early detection and treatment of cardiac arrhythmias has increased the adoption of CRM devices among patients and healthcare providers. Screening programs, patient education initiatives, and advocacy efforts have helped raise awareness about the importance of regular heart rhythm monitoring and timely intervention, driving market growth.
Furthermore, favorable reimbursement policies and healthcare infrastructure developments have facilitated greater access to CRM devices, particularly in emerging markets. Governments and private payers are increasingly recognizing the value of preventive care and investing in cardiovascular health initiatives, leading to broader adoption of CRM technologies globally.
Region Insights:
The CRM Devices Market exhibits regional variations in terms of market size, growth drivers, and regulatory landscape. North America dominates the market, driven by a high prevalence of cardiovascular diseases, well-established healthcare infrastructure, and favorable reimbursement policies. The United States accounts for the largest share of the North American market, supported by robust research and development activities, technological innovation, and a large patient pool.
Europe is another significant market for CRM devices, characterized by a growing elderly population and increasing incidence of cardiovascular diseases. Countries such as Germany, France, and the United Kingdom are major contributors to the European market, benefiting from advanced healthcare systems, strong regulatory frameworks, and widespread adoption of innovative medical technologies.
The Asia Pacific region is poised for substantial growth in the CRM Devices Market, fueled by rapid urbanization, changing lifestyles, and improving healthcare infrastructure. Countries like China, India, and Japan represent key growth opportunities, driven by rising healthcare expenditure, expanding middle-class population, and increasing awareness about cardiovascular health.
Latin America and the Middle East & Africa are emerging markets for CRM devices, characterized by growing healthcare spending, improving access to medical services, and rising prevalence of cardiovascular risk factors. While these regions face challenges such as limited healthcare resources and infrastructure gaps, increasing investments in healthcare infrastructure and government initiatives to improve access to healthcare services are expected to drive market growth.
Cardiac Rhythm Management Devices Market Scope
| Report Coverage | Details |
| Growth Rate from 2024 to 2033 | CAGR of 6.24% |
| Global Market Size in 2023 | USD 21.15 Billion |
| Global Market Size by 2033 | USD 38.75 Billion |
| U.S. Market Size in 2023 | USD 6.35 Billion |
| U.S. Market Size by 2033 | USD 11.36 Billion |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Segments Covered | By Product, By Application, and By End-use |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Cardiac Rhythm Management Devices Market Dynamics
Drivers:
Several factors are driving the growth of the CRM Devices Market. Firstly, the increasing prevalence of cardiovascular diseases, including atrial fibrillation, heart failure, and cardiac arrest, is a major driver of market growth. As the global burden of cardiovascular diseases continues to rise, there is a growing need for effective diagnostic and therapeutic solutions to manage these conditions.
Technological advancements in CRM devices have led to the development of more advanced and innovative products with improved accuracy, reliability, and patient outcomes. Miniaturization of devices, wireless connectivity, and remote monitoring capabilities have enhanced the usability and convenience of CRM devices, driving greater adoption among patients and healthcare providers.
Moreover, the growing emphasis on preventive care and early detection of cardiac arrhythmias has increased the demand for CRM devices. Screening programs, public awareness campaigns, and initiatives to promote heart health have encouraged individuals to undergo regular heart rhythm monitoring and seek timely medical intervention, boosting market growth.
Additionally, favorable reimbursement policies and healthcare reforms have expanded access to CRM devices, particularly in developed countries. Government initiatives to improve healthcare infrastructure, increase healthcare spending, and enhance reimbursement coverage for cardiac procedures have contributed to market growth by reducing financial barriers to treatment.
Furthermore, the aging population and increasing prevalence of risk factors such as obesity, diabetes, and hypertension have fueled the demand for CRM devices. As the elderly population grows, the incidence of cardiovascular diseases is expected to rise, driving greater adoption of diagnostic and therapeutic solutions for cardiac arrhythmias.
Opportunities:
The CRM Devices Market presents several opportunities for growth and innovation. Firstly, expanding indications for CRM devices and expanding the target patient population offer significant growth opportunities for manufacturers. The development of novel therapies for conditions such as atrial fibrillation, heart failure, and sudden cardiac arrest expands the addressable market for CRM devices, driving demand for innovative diagnostic and therapeutic solutions.
Moreover, technological advancements such as wearable devices, smartphone apps, and cloud-based platforms offer new avenues for remote monitoring and patient engagement. By leveraging digital health technologies, manufacturers can develop integrated solutions that provide real-time data insights, enable personalized patient care, and improve treatment outcomes.
Additionally, market expansion in emerging economies presents lucrative opportunities for growth. Rapid urbanization, increasing healthcare expenditure, and improving healthcare infrastructure in countries such as China, India, and Brazil create a favorable environment for market penetration and adoption of CRM devices.
Furthermore, partnerships and collaborations between manufacturers, healthcare providers, and research institutions can drive innovation and accelerate market growth. By pooling resources, expertise, and networks, stakeholders can collaborate on product development, clinical research, and market expansion initiatives, leading to the introduction of new products and services and increasing market penetration.
Challenges:
Despite the growth opportunities, the CRM Devices Market faces several challenges that may impede market growth. Firstly, regulatory complexities and approval processes can hinder product development and market entry, particularly for innovative technologies and novel therapies. Manufacturers must navigate stringent regulatory requirements and demonstrate safety, efficacy, and clinical utility to obtain regulatory approval for their products.
Moreover, reimbursement challenges and pricing pressures pose significant barriers to market access and adoption. Healthcare payers are increasingly scrutinizing the cost-effectiveness of medical devices and imposing stricter reimbursement policies, limiting reimbursement coverage for CRM procedures and reducing profitability for manufacturers.
Additionally, market competition and consolidation can impact market dynamics and intensify pricing pressures. The CRM Devices Market is highly competitive, with several major players competing for market share through product innovation, pricing strategies, and market expansion initiatives. Consolidation within the industry, through mergers and acquisitions, can lead to market concentration and reduce opportunities for smaller players.
Furthermore, technological complexities and interoperability issues can pose challenges to seamless integration and adoption of CRM devices within existing healthcare systems. Compatibility issues between different devices, software platforms, and electronic health record systems can hinder interoperability and data exchange, limiting the effectiveness of remote monitoring and patient management solutions.
Moreover, patient adherence and compliance with CRM therapy can impact treatment outcomes and healthcare costs. Patient education, counseling, and support programs are essential to promote treatment adherence and ensure optimal device utilization. However, barriers such as patient reluctance, lack of awareness, and socioeconomic factors can hinder patient engagement and adherence to therapy.
Read Also: Ammonium Chloride Market Size To Attain USD 2.24 Bn by 2033
Recent Developments
- In June 2023, Philips and BIOTRONIK formed a strategic alliance to expand care for out-of-hospital cardiology labs.
- In October 2023, MicroPort CRM launched the ULYS ICD and INVICTA Defibrillation Lead in Japan.
- In October 2023, Boston Scientific introduced the LUX-Dx II+ Insertable Cardiac Monitor System.
Cardiac Rhythm Management Devices Market Companies
- Physio-Control, Inc. (Stryker)
- BIOTRONIK
- Schiller
- Medtronic
- Abbott
- Koninklijke Philips N.V.
- Zoll Medical Corporation
- Schiller
- Boston Scientific Corporation
- Progetti Srl
- LivaNova Plc
Segments Covered in the Report
By Product
- Pacemakers
- Implantable
- External
- Defibrillators
- Implantable Cardioverter Defibrillators (ICD)
- S-ICD
- T-ICD
- Implantable Cardioverter Defibrillators (ICD)
- External Defibrillator
- Manual External Defibrillator
- Automatic External Defibrillator
- Wearable Cardioverter Defibrillator
- Cardiac Resynchronization Therapy (CRT)
- CRT-Defibrillator
- CRT-Pacemakers
By Application
- Congestive Heart Failure
- Arrhythmias
- Bradycardia
- Tachycardia
- Others
By End-use
- Hospitals
- Cardiac care centers
- Ambulatory surgical centers
- Others
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.uswebwire.com/
