Global Alzheimer’s therapeutics market poised for rapid expansion driven by rising disease prevalence and breakthrough therapies
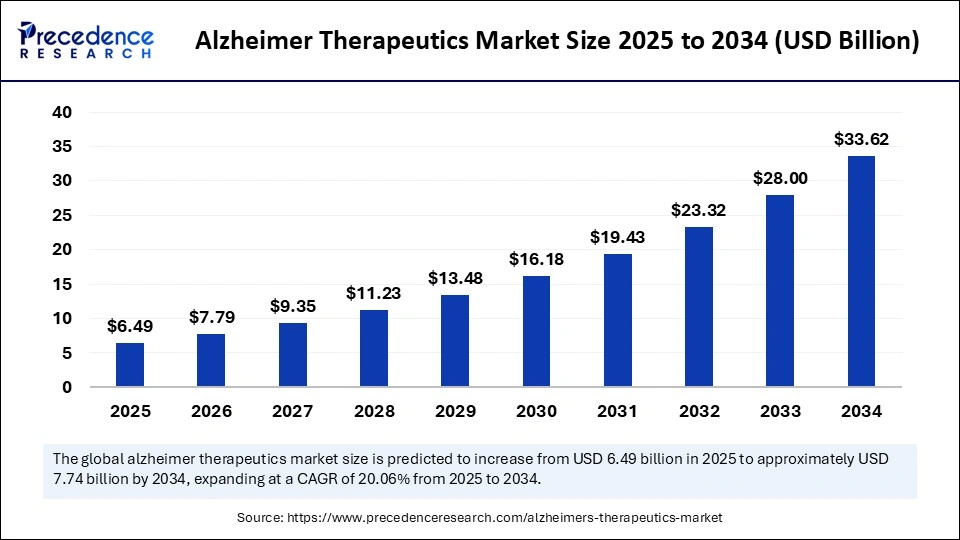
The global Alzheimer therapeutics market size is projected to experience robust growth, with expected market valuation hitting USD 33.62 billion by 2034 from USD 6.49 billion in 2025, growing at a CAGR of 20.06% between 2025 and 2034. This surge is primarily fueled by an increasing elderly population worldwide and revolutionary advances in disease-modifying therapies targeting Alzheimer’s pathology.
Alzheimer Therapeutics Market Key Points
-
The market value stood at USD 6.49 billion in 2025, forecasted to reach USD 33.62 billion by 2034, indicating strong growth momentum.
-
North America dominated with 46.10% market share in 2024, driven by extensive clinical research and healthcare infrastructure.
-
Asia Pacific is the fastest-growing region, expected to witness a CAGR of 10.90% from 2025–2034 due to rising geriatric population and government initiatives.
-
Cholinesterase inhibitors held the largest product segment share at 38.40% in 2024, widely used for mild to moderate Alzheimer’s treatment.
-
Anti-amyloid monoclonal antibodies are the fastest-growing segment, with a 12.80% CAGR, propelled by FDA approvals of drugs like Donanemab and Lecanemab.
-
Oral route of administration leads the market with 61.30% share in 2024, favored for convenience and cost-effectiveness.
-
Hospital pharmacies are the largest end-user segment with 44.90% market share, while online pharmacies are expanding rapidly at 11.80% CAGR.
Market Size and Growth Forecast (2025-2034)
| Year | Market Size (USD Billion) |
|---|---|
| 2025 | 6.49 |
| 2026 | 7.79 |
| 2034 | 33.62 |
| CAGR | 20.06% |
What Are Alzheimer Therapeutics?
Role of Artificial Intelligence in Alzheimer Therapeutics
AI is playing a pivotal role in accelerating Alzheimer’s therapeutics by enhancing diagnostic precision and speeding drug discovery. AI-driven neuroimaging tools improve early detection through better identification of brain changes indicative of Alzheimer’s onset. Furthermore, AI algorithms analyze vast datasets from clinical trials and real-world patient data to optimize therapeutic targets and personalize treatment plans. These innovations reduce the time and costs associated with drug development, supporting the rapid introduction of effective disease-modifying therapies.
What Factors Are Driving Market Growth?
The key drivers include the growing elderly population vulnerable to Alzheimer’s, substantial investments in R&D by pharmaceutical firms, and supportive healthcare policies promoting awareness and reimbursements. Increased government funding in clinical trials and innovative therapies also stimulates development. Advances in diagnostic technologies enable earlier detection, heightening demand for effective treatments. The rising prevalence of mild cognitive impairment (MCI) is creating new patient segments targeted by novel therapies, further boosting market expansion.
What Opportunities and Trends Are Emerging in Alzheimer Therapeutics?
How are personalized medicines impacting the market?
Personalized medicines tailored to an individual’s disease progression and genetic profile are gaining traction, leading to more effective treatments with fewer side effects. Companies are exploring biomarkers and genomic data to customize therapies, improving patient outcomes significantly.
What role do novel monoclonal antibodies play?
Monoclonal antibodies like Donanemab and Lecanemab, which clear amyloid beta plaques, represent a major breakthrough. Their approval is driving market growth by offering the first disease-modifying options that slow cognitive decline.
How is the online pharmacy segment influencing accessibility?
Online pharmacies are expanding rapidly by providing easier access to medications, especially for those in remote areas or with mobility challenges. Their growing presence ensures timely drug delivery and cost savings, encouraging patient adherence.
Regional and Segment Analysis
North America leads the Alzheimer therapeutics market, owning over 46% share due to abundant clinical trials and strong healthcare infrastructure.
The U.S. alone is projected to grow from USD 2.30 billion in 2025 to approximately USD 12.02 billion by 2034. Asia Pacific shows fastest expansion facilitated by government initiatives and growing disease awareness, with countries like China aggressively pursuing innovative therapies and national dementia action plans. Europe, Latin America, and the Middle East & Africa also contribute to global market growth as healthcare systems improve.
Segment-wise, cholinesterase inhibitors remain dominant in product type for symptom management, while anti-amyloid monoclonal antibodies lead in disease modification.
Mild to moderate Alzheimer’s disease patients constitute the largest stage-wise segment, with mild cognitive impairment segment growing rapidly.
Oral administration dominates for convenience, yet intravenous therapies are growing due to new FDA-approved monoclonal antibody treatments.
Hospital pharmacies are still the largest end-user, but online pharmacies are gaining momentum owing to patient convenience and digital adoption.
Latest Breakthroughs and Key Players
Major companies driving innovations include Biogen Inc., Eisai Co., Ltd., Eli Lilly & Company, Roche, Bristol Myers Squibb, Sanofi, and Novartis AG. Recent highlights include Eisai and Biogen’s announcement on continued benefits of lecanemab for early Alzheimer’s (July 2025), Roche’s advances in tau-targeting monoclonal antibodies and diagnostics presented at the AD/PD 2025 conference, and Sanofi’s collaboration with Vigil Neuroscience to bolster its early-stage pipeline. These breakthroughs underscore ongoing commitment to revolutionizing Alzheimer’s care.
Challenges and Cost Pressures
Despite promising advances, challenges like high drug development costs, prolonged clinical trials, and regulatory complexities persist. Furthermore, treatments such as monoclonal antibodies come with significant price tags, raising concerns about affordability and reimbursement.
Accessibility in emerging markets also remains limited due to infrastructure and economic constraints. Managing these hurdles requires continued investment and patient-centric policy frameworks.
Case Study Snapshot
Eisai’s collaboration with Biogen to bring lecanemab to market exemplifies a successful public-private partnership and clinical innovation. The antibody’s approval represents a milestone for disease-modifying Alzheimer’s treatments and offers hope for slowing disease progression in early-stage patients, illustrating the market’s potential impact when scientific breakthroughs are effectively translated into therapies.
Alzheimer Therapeutics Market Segment Covered in the Report
By Product Type
- Cholinesterase Inhibitors
- Donepezil
- Galantamine
- Rivastigmine
- NMDA Receptor Antagonist
- Cholinesterase + NMDA Combinations
- Anti-Amyloid Monoclonal Antibodies
- Others (Symptomatic Adjuncts)
By Stage of Alzheimers Disease
- Mild Cognitive Impairment due of Alzheimers
- Mild to Moderate Alzheimers Disease
- Moderate to Severe Alzheimers Disease
By Route of Administration
- Oral
- Transdermal
- Intravenous (IV)
By End User
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
Read Also: Incretin Mimetics Market
You can place an order or ask any questions. Please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344
