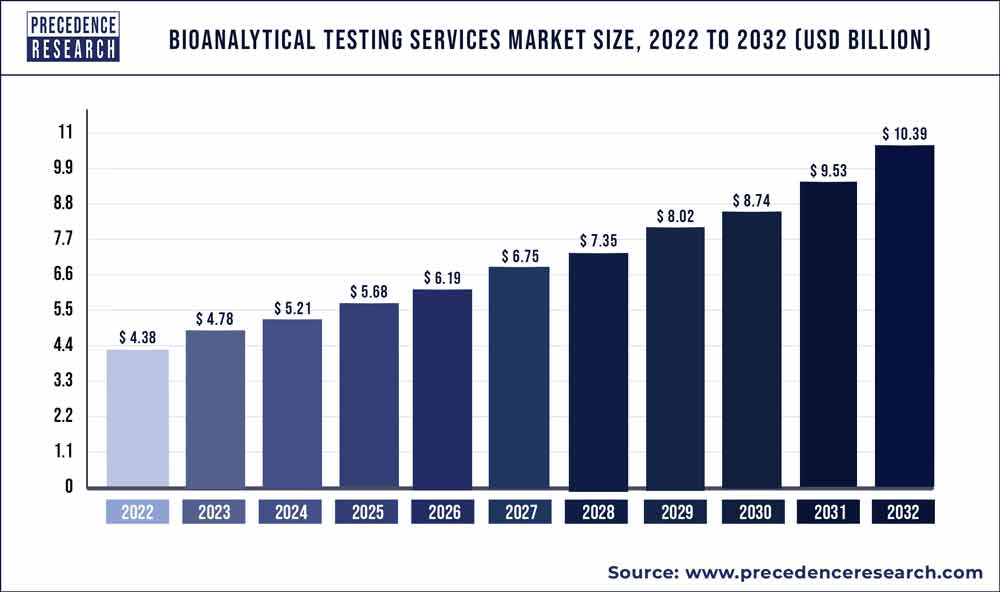
The bioanalytical testing services market size is poised to grow by USD 10.39 billion by 2032 from USD 4.38 billion in 2022, exhibiting a CAGR of 9.02% during the forecast period 2023-2032.
Key Takeaways:
- North America generated more than 47% of revenue share in 2022.
- By Test Type, the bioavailability segment contributed to the largest revenue share in 2022.
- By Application, the oncology segment is expected to expand at the fastest CAGR between 2023 and 2032.
Precedence Research has conducted a comprehensive market study that provides valuable insights into the performance of the market during the forecast period. The study identifies significant trends that are shaping the growth of the Bioanalytical testing services market. In this recently published report, essential dynamics such as drivers, restraints, and opportunities are highlighted for both established market players and emerging participants involved in production and supply.
To begin with, the Bioanalytical testing services Market report features an executive summary that offers a concise overview of the marketplace. It outlines the key players and industry categories expected to have an impact on the market in the coming years. The executive summary provides an unbiased summary of the market.
Get a Sample Report: https://www.precedenceresearch.com/sample/3107
Bioanalytical Testing Services Market Report Scope
| Report Coverage | Details |
| Market Size in 2023 | USD 4.78 Billion |
| Market Size by 2032 | USD 10.39 Billion |
| Growth Rate from 2023 to 2032 | CAGR of 9.02% |
| Largest Market | North America |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Segments Covered | By Test Type, By Molecules, By Application, and By End User |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Read More: Therapeutic Respiratory Devices Market Size To Rake USD 40.01 Bn By 2032
The empirical study on the global Bioanalytical testing services market primarily focuses on the drivers in subsequent sections. It demonstrates how changing demographics are projected to influence the supply and demand dynamics in the Bioanalytical testing services Market. Our market report for the Bioanalytical testing services market also delves into the significant rules and regulations that are likely to impact the future growth of this sector. Moreover, in order to comprehend the underlying demand factors, industry experts have provided insights into its fundamental origins.
Top Key Players:
- PPD, Inc.
- ICON Plc
- Charles River Laboratories International
- Covance, Inc.
- IQVIA
- Syneos Health
- SGS SA
- Toxikon
- Intertek Group Plc
- Pace Analytical Services LLC
Data Sources and Methodology
To gather comprehensive insights on the Global Bioanalytical testing services Market, we relied on a range of data sources and followed a well-defined methodology. Our approach involved interactions with industry experts and key stakeholders across the market’s value chain, including management organizations, processing organizations, and analytics service providers.
We followed a rigorous data analysis process to ensure the quality and credibility of our research. The gathered information was carefully evaluated, and relevant quantitative data was subjected to statistical analysis. By employing robust analytical techniques, we were able to derive meaningful insights and present a comprehensive overview of the Global Bioanalytical testing services Market.
The most resonating, simple, genuine, and important causes because of which you must decide to buy the Bioanalytical testing services market report exclusively from precedence research
- The research report has been meticulously crafted to provide comprehensive knowledge on essential marketing strategies and a holistic understanding of crucial marketing plans spanning the forecasted period from 2023 to 2032.
Key Features of the Report:
- Comprehensive Coverage: The report extensively encompasses a detailed explanation of highly effective analytical marketing methods applicable to companies across all industry sectors.
- Decision-Making Enhancement: It outlines a concise overview of the decision-making process while highlighting key techniques to enhance it, ensuring favorable business outcomes in the future.
- Articulated R&D Approach: The report presents a well-defined approach to conducting research and development (R&D) activities, enabling accurate data acquisition on current and future marketing conditions.
Bioanalytical Testing Services Market Segmentation:
By Test Type
- Pharmacokinetic
- Pharmacodynamics
- Bioavailability
- Bioequivalence
By Molecules
- Small Molecules
- Large Molecules
By Application
- Cardiology
- Gastroenterology
- Neurology
- Oncology
- Others
By End User
- Pharmaceutical and Biopharmaceutical Companies
- Contract Development and Manufacturing Organizations
- Contract Research Organizations
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Reasons to Consider Purchasing the Report:
- Enhance your market research capabilities by accessing this comprehensive and precise report on the global Bioanalytical testing services market.
- Gain a thorough understanding of the overall market landscape and be prepared to overcome challenges while ensuring robust growth.
- Benefit from in-depth research and analysis of the latest trends shaping the global Bioanalytical testing services market.
- Obtain detailed insights into evolving market trends, current and future technologies, and strategic approaches employed by key players in the global Bioanalytical testing services market.
- Receive valuable recommendations and guidance for both new entrants and established players seeking further market expansion.
- Discover not only the cutting-edge technological advancements in the global Bioanalytical testing services market but also the strategic plans of industry leaders.
Table of Content
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology (Premium Insights)
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Bioanalytical Testing Services Market
5.1. COVID-19 Landscape: Bioanalytical Testing Services Industry Impact
5.2. COVID 19 – Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Bioanalytical Testing Services Market, By Test Type
8.1. Bioanalytical Testing Services Market, by Test Type, 2023-2032
8.1.1. Pharmacokinetic
8.1.1.1. Market Revenue and Forecast (2020-2032)
8.1.2. Pharmacodynamics
8.1.2.1. Market Revenue and Forecast (2020-2032)
8.1.3. Bioavailability
8.1.3.1. Market Revenue and Forecast (2020-2032)
8.1.4. Bioequivalence
8.1.4.1. Market Revenue and Forecast (2020-2032)
Chapter 9. Global Bioanalytical Testing Services Market, By Molecules
9.1. Bioanalytical Testing Services Market, by Molecules, 2023-2032
9.1.1. Small Molecules
9.1.1.1. Market Revenue and Forecast (2020-2032)
9.1.2. Large Molecules
9.1.2.1. Market Revenue and Forecast (2020-2032)
Chapter 10. Global Bioanalytical Testing Services Market, By Application
10.1. Bioanalytical Testing Services Market, by Application, 2023-2032
10.1.1. Cardiology
10.1.1.1. Market Revenue and Forecast (2020-2032)
10.1.2. Gastroenterology
10.1.2.1. Market Revenue and Forecast (2020-2032)
10.1.3. Neurology
10.1.3.1. Market Revenue and Forecast (2020-2032)
10.1.4. Oncology
10.1.4.1. Market Revenue and Forecast (2020-2032)
10.1.5. Others
10.1.5.1. Market Revenue and Forecast (2020-2032)
Chapter 11. Global Bioanalytical Testing Services Market, By End User
11.1. Bioanalytical Testing Services Market, by End User, 2023-2032
11.1.1. Pharmaceutical and Biopharmaceutical Companies
11.1.1.1. Market Revenue and Forecast (2020-2032)
11.1.2. Contract Development and Manufacturing Organizations
11.1.2.1. Market Revenue and Forecast (2020-2032)
11.1.3. Contract Research Organizations
11.1.3.1. Market Revenue and Forecast (2020-2032)
Chapter 12. Global Bioanalytical Testing Services Market, Regional Estimates and Trend Forecast
12.1. North America
12.1.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.1.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.1.3. Market Revenue and Forecast, by Application (2020-2032)
12.1.4. Market Revenue and Forecast, by End User (2020-2032)
12.1.5. U.S.
12.1.5.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.1.5.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.1.5.3. Market Revenue and Forecast, by Application (2020-2032)
12.1.5.4. Market Revenue and Forecast, by End User (2020-2032)
12.1.6. Rest of North America
12.1.6.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.1.6.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.1.6.3. Market Revenue and Forecast, by Application (2020-2032)
12.1.6.4. Market Revenue and Forecast, by End User (2020-2032)
12.2. Europe
12.2.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.2.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.2.3. Market Revenue and Forecast, by Application (2020-2032)
12.2.4. Market Revenue and Forecast, by End User (2020-2032)
12.2.5. UK
12.2.5.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.2.5.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.2.5.3. Market Revenue and Forecast, by Application (2020-2032)
12.2.5.4. Market Revenue and Forecast, by End User (2020-2032)
12.2.6. Germany
12.2.6.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.2.6.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.2.6.3. Market Revenue and Forecast, by Application (2020-2032)
12.2.6.4. Market Revenue and Forecast, by End User (2020-2032)
12.2.7. France
12.2.7.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.2.7.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.2.7.3. Market Revenue and Forecast, by Application (2020-2032)
12.2.7.4. Market Revenue and Forecast, by End User (2020-2032)
12.2.8. Rest of Europe
12.2.8.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.2.8.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.2.8.3. Market Revenue and Forecast, by Application (2020-2032)
12.2.8.4. Market Revenue and Forecast, by End User (2020-2032)
12.3. APAC
12.3.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.3.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.3.3. Market Revenue and Forecast, by Application (2020-2032)
12.3.4. Market Revenue and Forecast, by End User (2020-2032)
12.3.5. India
12.3.5.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.3.5.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.3.5.3. Market Revenue and Forecast, by Application (2020-2032)
12.3.5.4. Market Revenue and Forecast, by End User (2020-2032)
12.3.6. China
12.3.6.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.3.6.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.3.6.3. Market Revenue and Forecast, by Application (2020-2032)
12.3.6.4. Market Revenue and Forecast, by End User (2020-2032)
12.3.7. Japan
12.3.7.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.3.7.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.3.7.3. Market Revenue and Forecast, by Application (2020-2032)
12.3.7.4. Market Revenue and Forecast, by End User (2020-2032)
12.3.8. Rest of APAC
12.3.8.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.3.8.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.3.8.3. Market Revenue and Forecast, by Application (2020-2032)
12.3.8.4. Market Revenue and Forecast, by End User (2020-2032)
12.4. MEA
12.4.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.4.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.4.3. Market Revenue and Forecast, by Application (2020-2032)
12.4.4. Market Revenue and Forecast, by End User (2020-2032)
12.4.5. GCC
12.4.5.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.4.5.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.4.5.3. Market Revenue and Forecast, by Application (2020-2032)
12.4.5.4. Market Revenue and Forecast, by End User (2020-2032)
12.4.6. North Africa
12.4.6.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.4.6.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.4.6.3. Market Revenue and Forecast, by Application (2020-2032)
12.4.6.4. Market Revenue and Forecast, by End User (2020-2032)
12.4.7. South Africa
12.4.7.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.4.7.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.4.7.3. Market Revenue and Forecast, by Application (2020-2032)
12.4.7.4. Market Revenue and Forecast, by End User (2020-2032)
12.4.8. Rest of MEA
12.4.8.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.4.8.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.4.8.3. Market Revenue and Forecast, by Application (2020-2032)
12.4.8.4. Market Revenue and Forecast, by End User (2020-2032)
12.5. Latin America
12.5.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.5.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.5.3. Market Revenue and Forecast, by Application (2020-2032)
12.5.4. Market Revenue and Forecast, by End User (2020-2032)
12.5.5. Brazil
12.5.5.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.5.5.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.5.5.3. Market Revenue and Forecast, by Application (2020-2032)
12.5.5.4. Market Revenue and Forecast, by End User (2020-2032)
12.5.6. Rest of LATAM
12.5.6.1. Market Revenue and Forecast, by Test Type (2020-2032)
12.5.6.2. Market Revenue and Forecast, by Molecules (2020-2032)
12.5.6.3. Market Revenue and Forecast, by Application (2020-2032)
12.5.6.4. Market Revenue and Forecast, by End User (2020-2032)
Chapter 13. Company Profiles
13.1. PPD, Inc.
13.1.1. Company Overview
13.1.2. Product Offerings
13.1.3. Financial Performance
13.1.4. Recent Initiatives
13.2. ICON Plc
13.2.1. Company Overview
13.2.2. Product Offerings
13.2.3. Financial Performance
13.2.4. Recent Initiatives
13.3. Charles River Laboratories International
13.3.1. Company Overview
13.3.2. Product Offerings
13.3.3. Financial Performance
13.3.4. Recent Initiatives
13.4. Covance, Inc.
13.4.1. Company Overview
13.4.2. Product Offerings
13.4.3. Financial Performance
13.4.4. Recent Initiatives
13.5. IQVIA
13.5.1. Company Overview
13.5.2. Product Offerings
13.5.3. Financial Performance
13.5.4. Recent Initiatives
13.6. Syneos Health
13.6.1. Company Overview
13.6.2. Product Offerings
13.6.3. Financial Performance
13.6.4. Recent Initiatives
13.7. SGS SA
13.7.1. Company Overview
13.7.2. Product Offerings
13.7.3. Financial Performance
13.7.4. Recent Initiatives
13.8. Toxikon
13.8.1. Company Overview
13.8.2. Product Offerings
13.8.3. Financial Performance
13.8.4. Recent Initiatives
13.9. Intertek Group Plc
13.9.1. Company Overview
13.9.2. Product Offerings
13.9.3. Financial Performance
13.9.4. Recent Initiatives
13.10. Pace Analytical Services LLC
13.10.1. Company Overview
13.10.2. Product Offerings
13.10.3. Financial Performance
13.10.4. Recent Initiatives
Chapter 14. Research Methodology
14.1. Primary Research
14.2. Secondary Research
14.3. Assumptions
Chapter 15. Appendix
15.1. About Us
15.2. Glossary of Terms
About Us:
Our team comprises a dedicated group of research analysts and management consultants who are driven by a unified vision: assisting individuals and organizations in realizing their strategic objectives, both immediate and long-term, through the provision of comprehensive research services. At Precedence Research, we have positioned ourselves to cater to the needs of a diverse range of entities, including established companies, startups, and non-profit organizations across various sectors. Our expertise extends to industries such as packaging, automotive, healthcare, chemicals and materials, industrial automation, consumer products, electronics and semiconductors, IT and telecommunications, and energy. With a wealth of experience within our ranks, our skilled analysts are equipped with extensive knowledge of the research landscape.
Contact Us
Precedence Research
Apt 1408 1785 Riverside Drive Ottawa, ON, K1G 3T7, Canada
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
